VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00129
1233335-78-8
C₂₀H₁₇FO₆
372.34

VLIM-00130
1987957-28-7
C₂₈H₆₁FN₅O₁₄P₃
803.73

VLIM-00131
103532-27-0
C₂₂H₂₆N₂O₄S
414.52
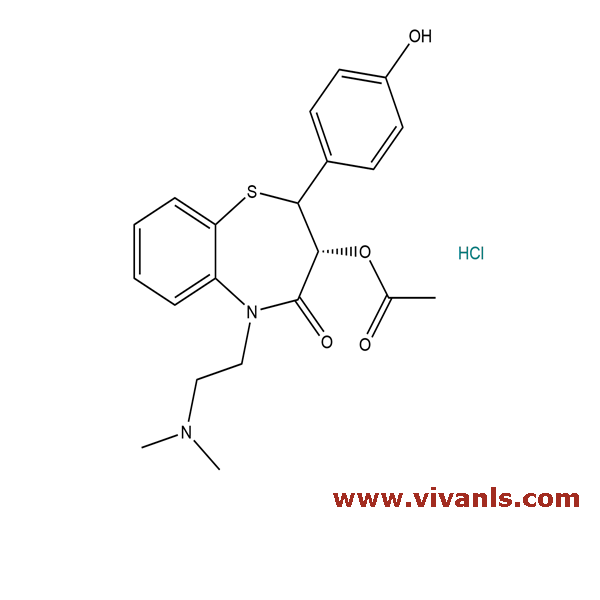
VLIM-00132
142926-07-6
C₂₁H₂₅ClN₂O₄S
436.95

VLIM-00133
86408-44-8
C₁₉H₂₂N₂O₃S.HCl
394.91

VLIM-00134
115992-91-1 (base)
C₁₇H₁₈N₂O₃S
330.4

VLIM-00135
88150-42-9
C₂₀H₂₅ClN₂O₅
408.88

VLIM-00136
88150-62-3
C₂₅H₃₄ClNO₆
480.01

VLIM-00137
173532-15-5
C₁₁H₁₀ClN₅OS
295.75

VLIM-00138
583840-03-3
C₂₁H₁₆ClF₃N₄O₄
480.82
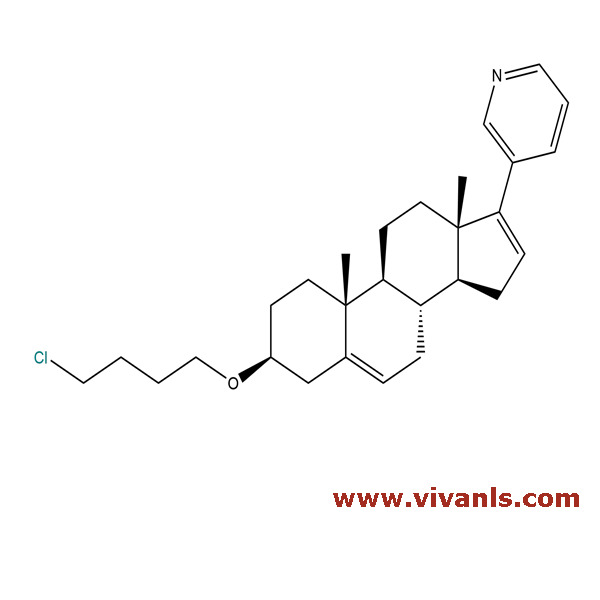
VLIM-00139
NA
C₂₁H₃₀O₃
330.47

VLIM-00140
1296129-15-1
C₂₄H₂₁F₂NO₃
409.44