VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
propionamide-1722927117.png)
VLIM-01082
2759-54-8
C9H10ClNO
183.64
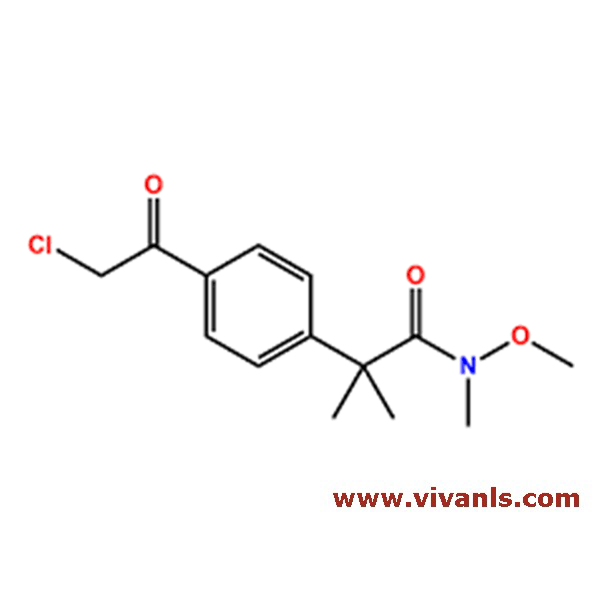
VLIM-01083
1638785-15-5
C28H35N3O4
477.6

VLIM-01084
2099128-83-1
C24H27N3O3
405.49

VLIM-01085
96382-71-7
C18H17Cl2NO4
382.2

VLIM-01086
101666-68-6
C37H67NO13
733.93
![Impurities-SACUBITRIL impurity [ Delta B4 (Ester Impurity) ]-1724146620.png](/images/productimages/Impurities-SACUBITRIL impurity [ Delta B4 (Ester Impurity) ]-1724146620.png)
VLIM-01087
2131788-25-3
C25H31NO4
409.5
![Impurities-SACUBITRIL impurity [Delta C1 Racemic Mixture ]-1724146666.png](/images/productimages/Impurities-SACUBITRIL impurity [Delta C1 Racemic Mixture ]-1724146666.png)
VLIM-01088
1012341-48-8
C23H27NO4
381.47

VLIM-01089
174607-70-6
C21H27NO4
357.5

VLIM-01090
27475-14-5
C17H15BrO4
363.2

VLIM-01091
960404-86-8
C21H25ClO7
424.9

VLIM-01092
63295-95-4
C15H14O4
244.3

VLIM-01093
1620323-43-4
C27H35NO2
405.6