VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-01033
73851-70-4
C₁₃H₂₂N₄O₄S
330.4

VLIM-01034
117846-02-3
C₁₂H₁₉N₃O₄S
301.4

VLIM-01035
207592-21-0
C₂₇H₄₄N₈O₆S₂
640.8

VLIM-01036
73857-20-2
C₁₃H₂₂N₄O₄S
330.4

VLIM-01037
104632-27-1
C₁₀H₁₉Cl₂N₃S
284.25

VLIM-01038
931-86-2
C₃H₄N₄O
112.09

VLIM-01039
2169998-23-4
C₂₁H₂₃ClO₇
422.86
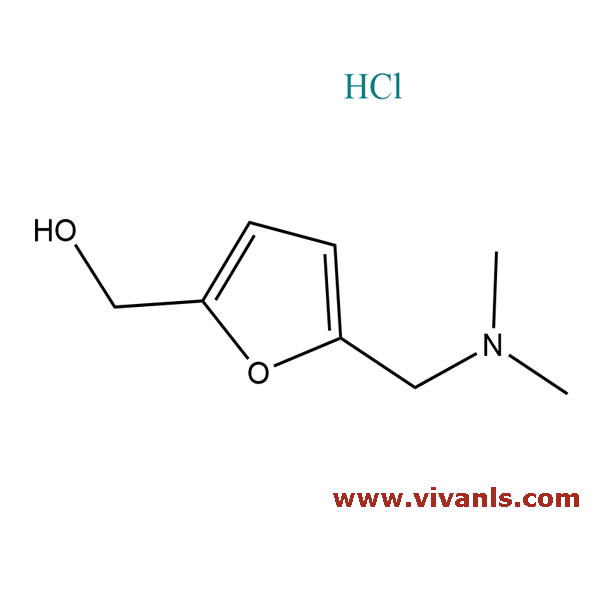
VLIM-01040
81074-81-9
C₈H₁₄ClNO₂
191.66

VLIM-01041
72078-82-1
C₃H₆N₂O₃
118.09

VLIM-01042
172152-37-3
C₁₉H₁₈N₄O₃S
382.44

VLIM-01043
41806-25-1
C₁₄H₁₁ClO₂
246.69

VLIM-01044
147663-30-7
C₂₆H₄₀N₂O₅
460.6