VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
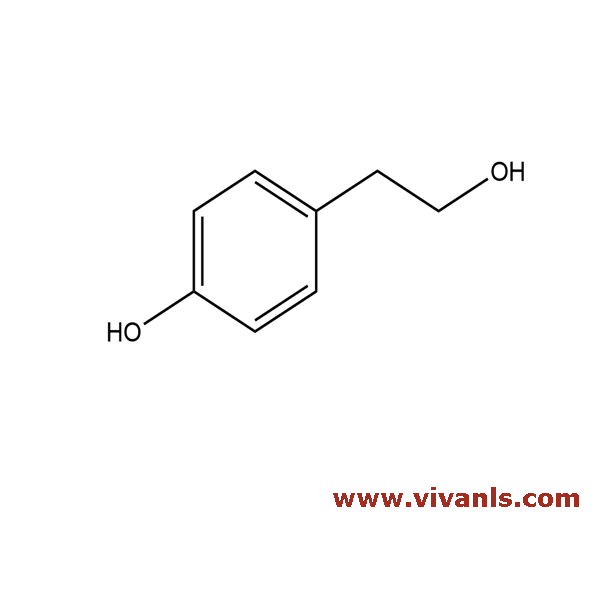
VLIM-01058
501-94-0
C₈H₁₀O₂
138.16

VLIM-01059
912570-13-9
C12H10BrNO
264.1

VLIM-01060
377756-15-5
C12H10BrNO
264.1

VLIM-01061
NA
C14H23NO3
253.34

VLIM-01062
1686124-74-2
C16H23N3O6
353.37

VLIM-01063
1189889-44-8
C8H10N2O2
166.2
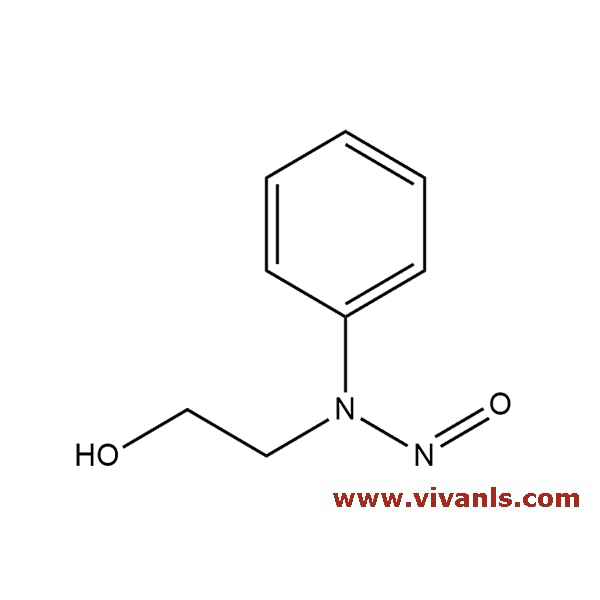
VLIM-01064
25413-76-7
C8H10N2O2
166.2

VLIM-01065
NA
C21H20N4O6
424.4

VLIM-01066
NA
C13H16ClN3O4
313.7

VLIM-01067
124832-31-1
C21H26N6O6
458.5

VLIM-01068
101666-68-6
C37H67NO13
733.93

VLIM-01069
1797879-37-8
C26H29N3O
399.54