VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-01045
2100872-88-4
C₂₁H₂₇ClO₇
426.9

VLIM-01047
99-06-9
HOC₆H₄CO₂H
138.12

VLIM-01048
51-36-5
Cl₂C₆H₃CO₂H
191.01

VLIM-01049
619-14-7
HOC₆H₃(NO₂)CO₂H
183.12

VLIM-01050
66622-47-7
C₁₃H₁₈O₂
206.28

VLIM-01051
3585-52-2
C₁₁H₁₄O₂
178.23

VLIM-01052
96948-64-0
C₁₂H₁₉NO₂
209.28
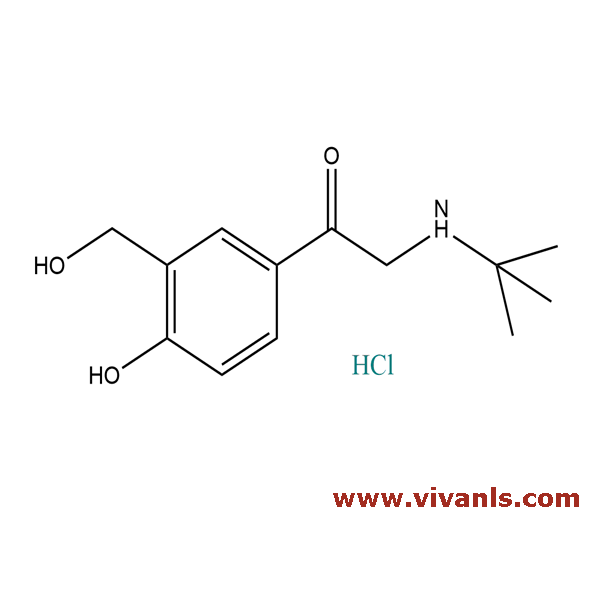
VLIM-01053
41489-89-8
C₁₃H₂₀ClNO₃
273.76

VLIM-01054
25162-00-9
C₁₀H₁₄N₂
162.23

VLIM-01055
1062122-63-7
C₅₃H₈₃NO₁₄
958.22

VLIM-01056
1708118-13-1
C₅₃H₈₃NO₁₄
958.22

VLIM-01057
112233-23-5
C₅H₉N₃OS : HCl
195.7