VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-01008
3070-86-8
C₁₆H₁₆N₂O₃
284.31

VLIM-01009
948551-71-1
C₂₇H₂₈N₆O₄
500.55
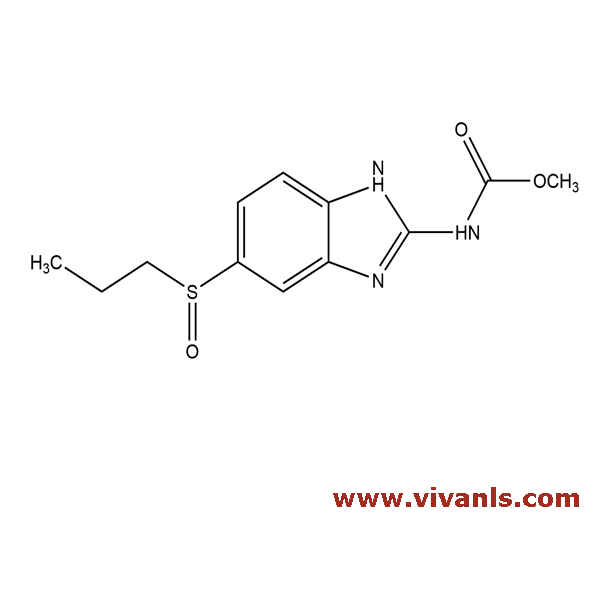
VLIM-01010
54029-12-8
C₁₂H₁₅N₃O₃S
281.33

VLIM-01011
34523-34-7
C₈H₉NO₂
151.16

VLIM-01012
99-93-4
C₈H₈O₂
136.15

VLIM-01013
2623-33-8
C₁₀H₁₁NO₃
193.2

VLIM-01014
103-84-4
C₈H₉NO
135.16

VLIM-01015
614-80-2
C₈H₉NO₂
151.16

VLIM-01016
118-93-4
C₈H₈O₂
136.15

VLIM-01017
152943-33-4
C₉H₉ClO₂
184.62
-1684476701.png)
VLIM-01018
148553-51-9
C₈H₁₇NO₂
159.23
-1684477420.png)
VLIM-01019
181289-15-6
C₉H₁₇NO₃
187.24