VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
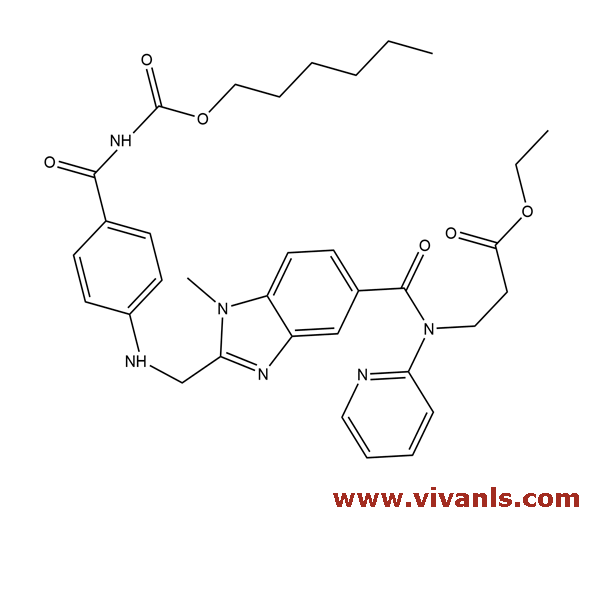
VLIM-01020
1580491-16-2
C₃₂H₃₈N₈O₄
598.7

VLIM-01021
211914-96-4
C₂₉H₃₁N₇O₅
557.61

VLIM-01022
N/A
C₃₀H₃₃N₇O₅
571.63

VLIM-01023
34911-51-8
C₉H₈BrClO
247.52
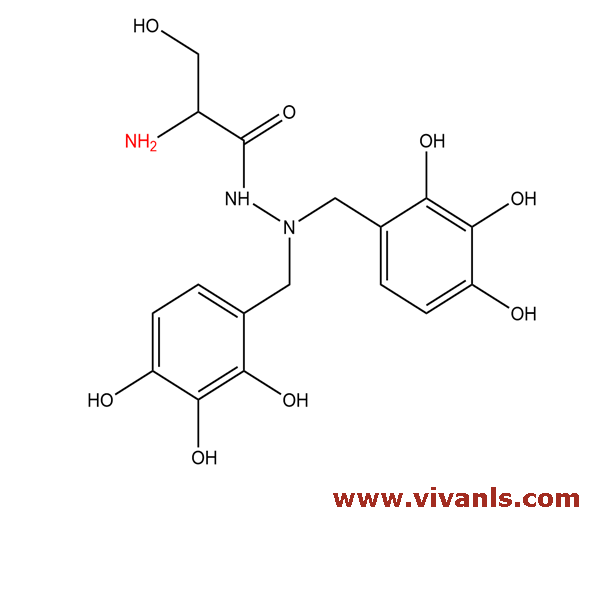
VLIM-01024
2472968-83-3
C₁₇H₂₁N₃O₈
395.4

VLIM-01025
784143-42-6
C₁₈H₂₁N₃O₃S
359.44

VLIM-01026
2213398-75-3
C₁₈H₁₅N₅O
317.4

VLIM-01028
6974-32-9
C₂₈H₂₄O₉
504.48

VLIM-01029
54030-32-9
C₈H₈O₃
152.15

VLIM-01030
1789703-37-2
C₁₇H₂₅N₃O₂
303.41

VLIM-01031
72126-78-4
C₂₂H₃₅N₅O₄S₂
497.67

VLIM-01032
66356-53-4
C₁₀H₁₈N₂OS
214.33