VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00996
7640-51-9
C₁₇H₂₀N₂OS
300.42
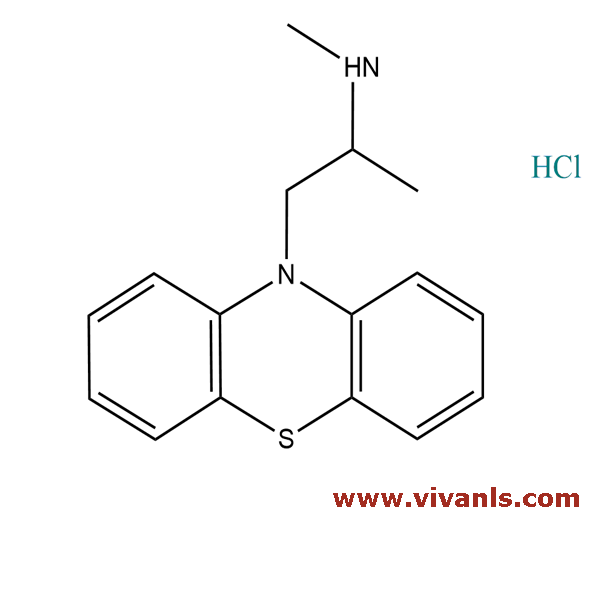
VLIM-00997
60113-77-1
C₁₆H₁₉ClN₂S
270.39

VLIM-00998
117796-52-8
C₂₁H₂₁ClN₂O
352.86
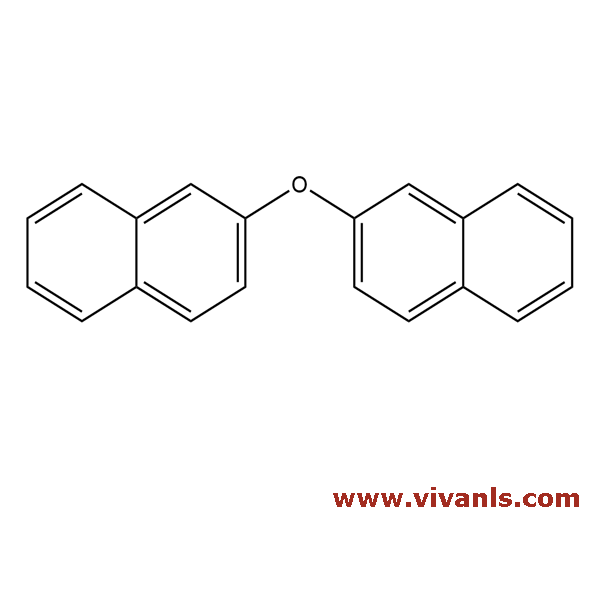
VLIM-00999
613-80-9
C₂₀H₁₄O
270.33

VLIM-01000
3964-54-3
C₈H₈ClNO₂
185.61

VLIM-01001
1693-37-4
C₉H₁₁NO₂
165.19

VLIM-01002
612069-28-0
C₃₈H₇₀N₂O₁₃
762.97

VLIM-01003
123967-58-8
C₃₉H₇₁NO₁₃
761.98
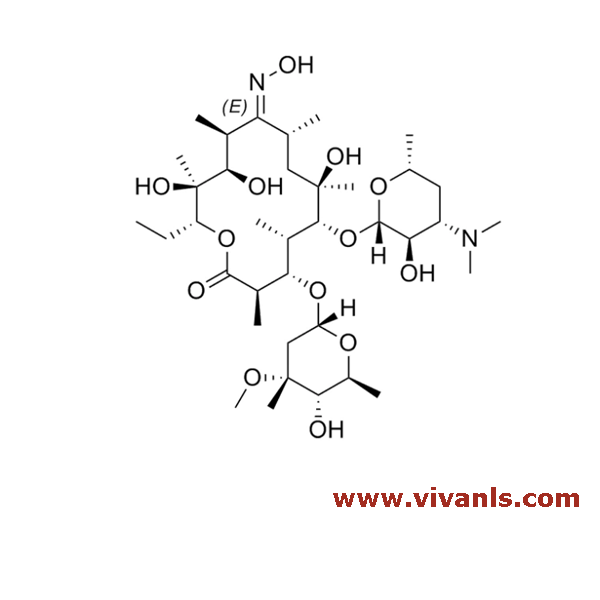
VLIM-01004
105900-46-7
C₃₇H₆₇NO₁₃
733.93

VLIM-01005
128940-83-0
C₃₉H₇₁NO₁₃
762

VLIM-01006
56-12-2
NH₂(CH₂)₃COOH
103.12
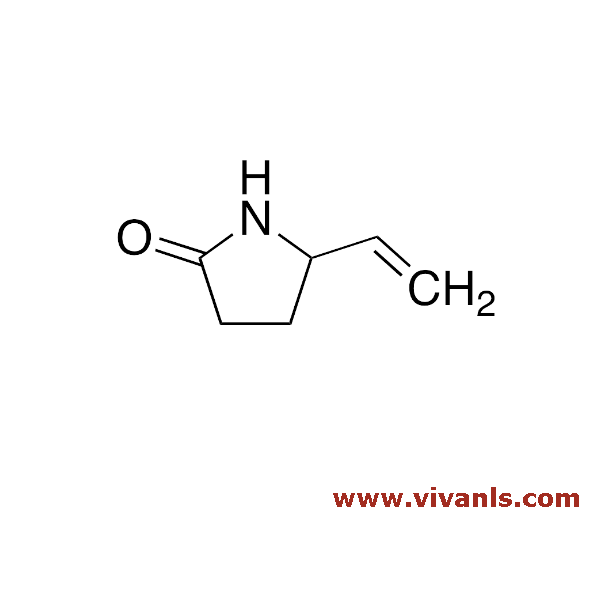
VLIM-01007
7529-16-0
C₆H₉NO
111.14