VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00960
538-71-6
C₂₂H₄₀BrNO
414.46

VLIM-00961
213476-12-1
C₁₆H₁₄F₃N₃O₃S
385.36

VLIM-00962
102767-31-7
C₈H₁₅ClN₂O₂
206.67

VLIM-00963
490-79-9
(HO)₂C₆H₃CO₂H
154.12
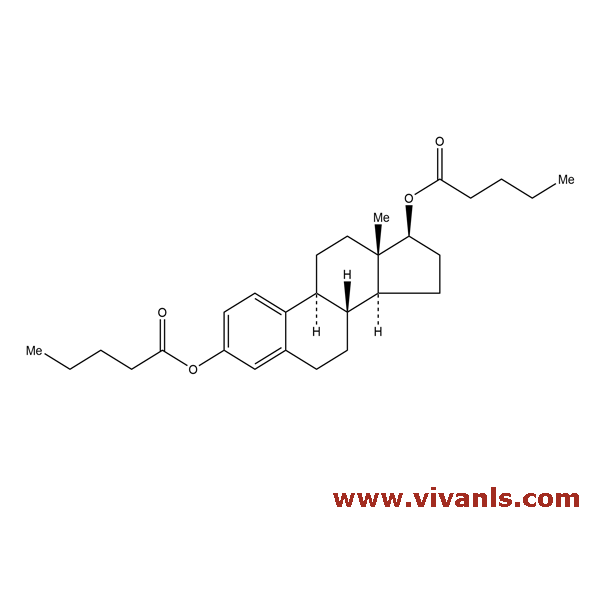
VLIM-00964
63042-28-4
C₂₈H₄₀O₄
440.61

VLIM-00965
21881-45-8
C₂₃H₃₂O₃
356.5

VLIM-00966
4921-49-7
C₈H₉ClN₄O₂
228.64

VLIM-00967
1887057-05-7
C₄₁H₅₅NO₁₄
785.88

VLIM-00968
707-36-8
C₁₂H₁₉Cl
198.73

VLIM-00969
1239692-16-0
C₁₁H₂₃NO₂
237.77

VLIM-00970
1909294-27-4
C₁₀H₂₁NO₂ . HCl
223.74

VLIM-00971
153381-68-1
C₄₃H₅₃NO₁₄
807.88