VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00924
2484719-11-9
C₂₆H₃₃NO₃
407.55
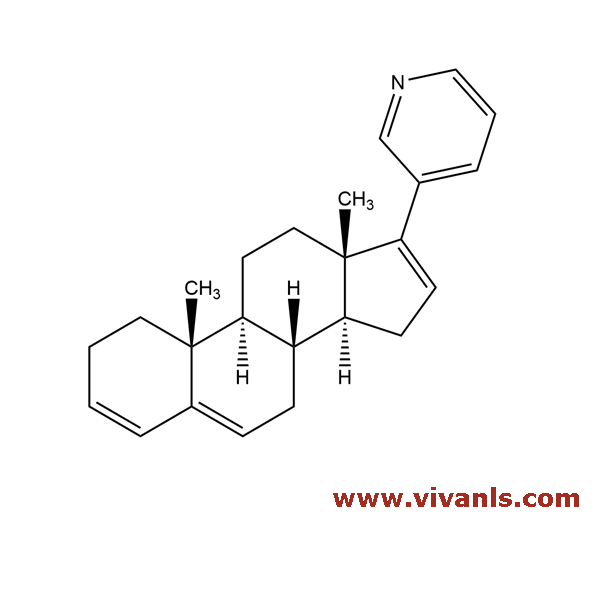
VLIM-00925
154229-20-6
C₂₄H₂₉N
331.49

VLIM-00926
81103-14-2
C₃₉H₇₁NO₁₃
761.98

VLIM-00927
1391194-45-8
C₂₀H₂₂ClNO₃S
391.91
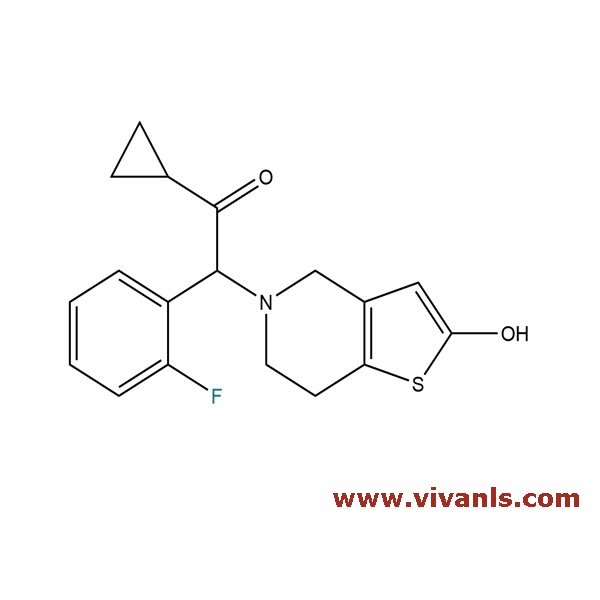
VLIM-00928
201049-74-3
C₁₈H₁₈FNO₂S
331.4
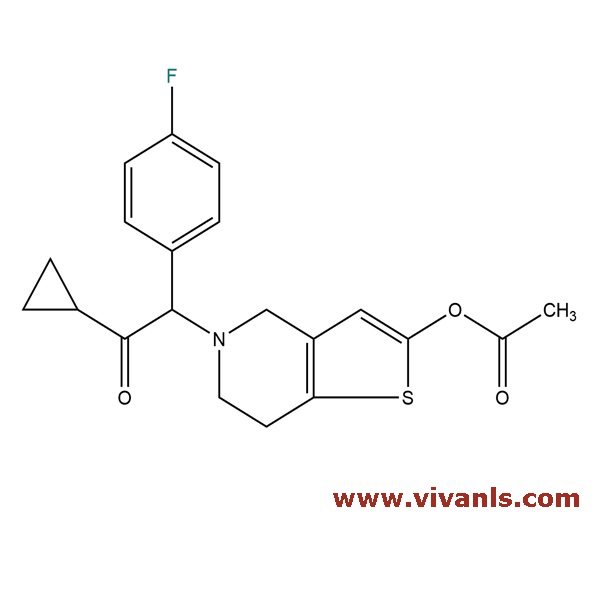
VLIM-00929
1391194-50-5
C₂₀H₂₀FNO₃S
373.44

VLIM-00930
408309-07-9
C₂₇H₃₃N
371.57

VLIM-00931
7240-38-2
C₁₉H₁₈N₃O₅SNa · H₂O
441.43
-1682503339.png)
VLIM-00932
581-96-4
C₁₂H₁₀O₂
186.21

VLIM-00933
121-71-1
C₈H₈O₂
136.15
![Impurities-3-Chloro-10,11-dihydro-5H-dibenzo[b,f]azepine-1682503807.png](/images/productimages/Impurities-3-Chloro-10,11-dihydro-5H-dibenzo[b,f]azepine-1682503807.png)
VLIM-00934
32943-25-2
C₁₄H₁₂ClN
229.711
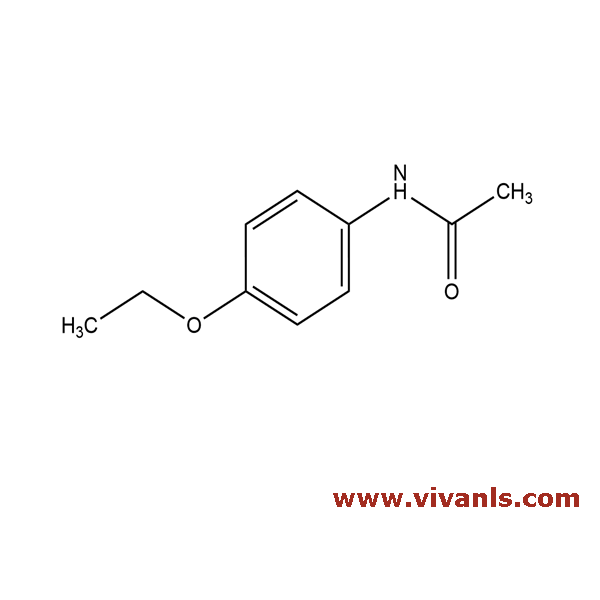
VLIM-00935
62-44-2
CH₃CONHC₆H₄OC₂H₅
179.22