VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00910
2484719-15-3
C₂₇H₃₇NO
391.59

VLIM-00911
2484719-17-5
C₂₈H₃₈ClNO
440.06

VLIM-00912
2410075-48-6
C₂₆H₃₁NO₃
405.53

VLIM-00913
50910-55-9
C₇H₅Br₂NO
278.9

VLIM-00914
NA
C₁₄H₁₀N₆O
158.17

VLIM-00915
NA
C₃₄H₄₀N₈O₆
656.73
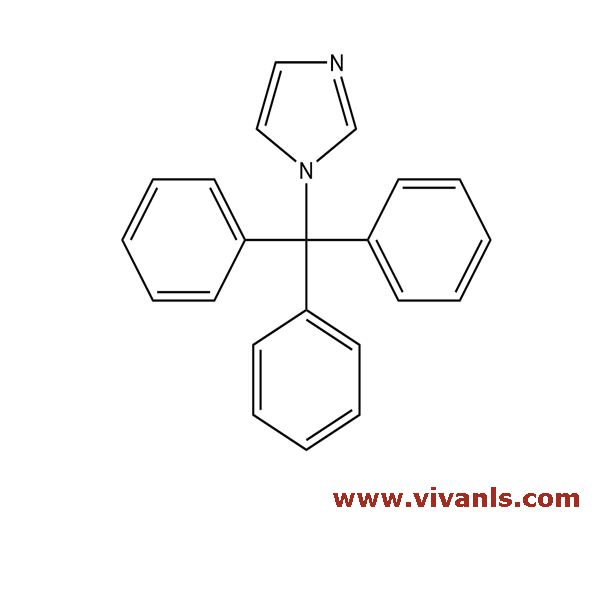
VLIM-00916
15469-97-3
C₂₂H₁₈N₂
310.4

VLIM-00917
2158184-11-1
C₂₇H₂₃ClN₄
438.95
-2-(ethylamino)propan-1-one hydrochloride-1681372447.png)
VLIM-00918
NA
C₁₁H₁₅Cl₂NO
248.19
amino)-1-(3-chlorophenyl) propan-1-one hydrochloride-1681372491.png)
VLIM-00919
1193779-78-0
C₁₄H₂₁Cl₂NO
290.23
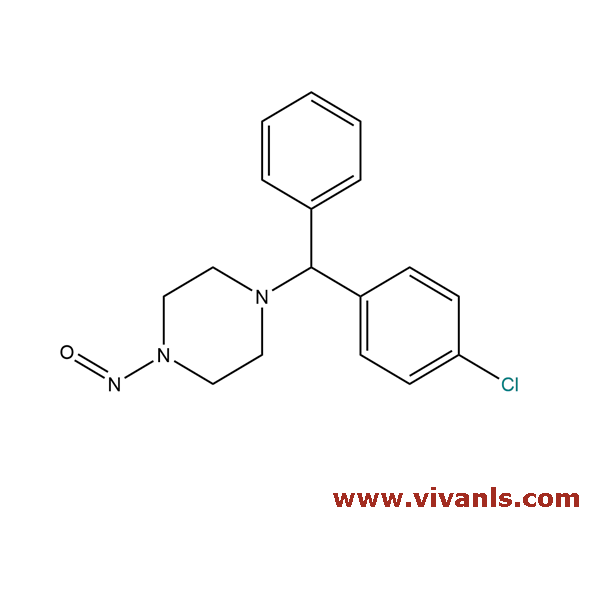
VLIM-00920
2005-04-01
C₁₇H₁₈ClN₃O
315.8
 propan-1-one-1681383904.png)
VLIM-00923
75815-22-4
C₉H₈BrClO
247.52