VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00885
1662702-90-0
C₁₅H₁₅ClO
246.73
-3,4-dimethoxypyridine hydrochloride-1666260886.png)
VLIM-00886
72830-09-2
C₈H₁₁Cl₂NO₂
224.08

VLIM-00887
89848-51-1
C₃₅H₃₈Cl₂N₈O₄
705.63

VLIM-00888
3023702-71-5
C₄₄H₄₁Cl₄N₁₁O₆
961.68

VLIM-00889
89848-49-7
C₃₄H₃₆Cl₂N₈O₄
691.61
 propan-1-one-1681364166.png)
VLIM-00890
877-37-2
C₉H₈BrClO
247.52
 propan-1-one-1681364888.png)
VLIM-00891
87427-61-0
C₉H₇BrCl₂O
281.96
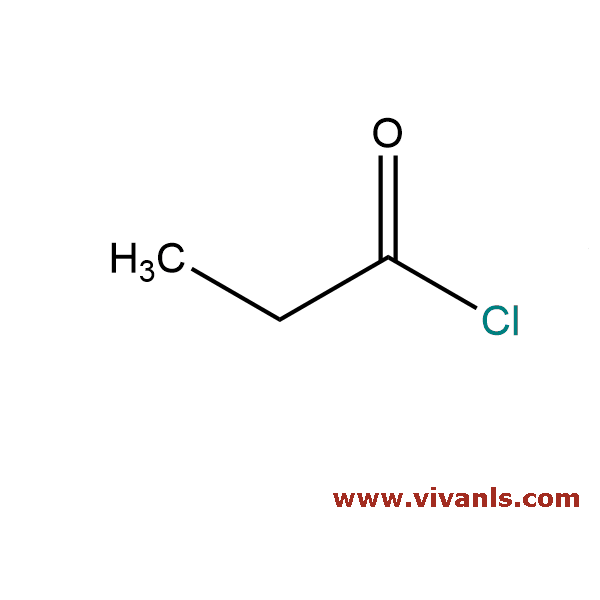
VLIM-00892
79-03-8
CH₃CH₂COCl
92.52

VLIM-00893
42399-49-5
C₁₆H₁₅NO₃S
301.36

VLIM-00894
50739-76-9
C₇H₇Br₂NO
280.94

VLIM-00895
NA
C₁₇H₂₄N₄O₃
332.4

VLIM-00897
5790-46-5
C₁₃H₂₁NO₂
223.31