VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00848
155748-61-1
C₁₆H₁₂Cl₂O
291.17

VLIM-00849
79645-15-1
C₁₇H₁₈Cl₃N
342.69

VLIM-00850
2731620-64-5
C₂₅H₄₀O₅
420.58

VLIM-00851
101314-97-0
C₂₅H₃₉NaO₆
458.6

VLIM-00852
145576-25-6
C₂₇H₄₀O₆
460.6

VLIM-00853
79902-42-4
C₂₃H₃₄O₅
390.5
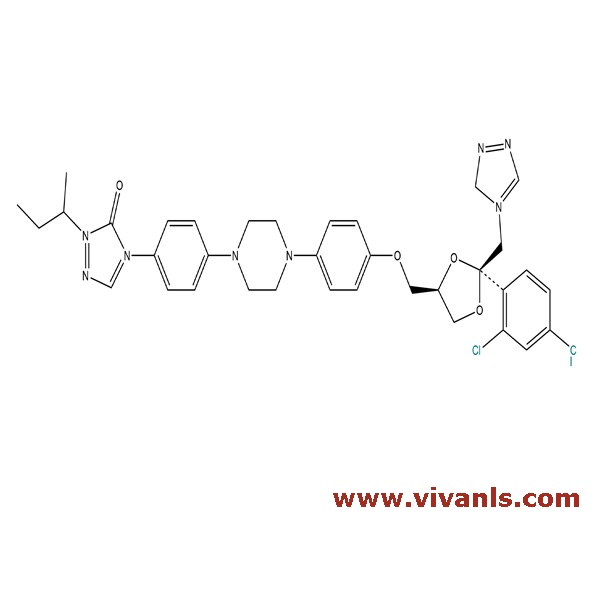
VLIM-00854
854372-77-3
C₃₅H₃₈Cl₂N₈O₄
705.63

VLIM-00855
1789703-37-2
C₁₇H₂₅N₃O₂
303.4

VLIM-00856
7597-60-6
C₇H₁₀N₄O₃
198.18

VLIM-00857
20041-90-1
C₇H₁₂N₄O
168.2

VLIM-00858
110-56-5
C₄H₈Cl₂
127.01
-1665661337.png)
VLIM-00859
461432-25-7
C₂₉H₃₃ClO₁₀
577.02