VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
-Isomer-1664368548.png)
VLIM-00810
2307668-79-5
C₂₅H₃₁NO₅
425.5
-Isomer-1664368834.png)
VLIM-00811
766480-48-2
C₂₄H₂₉NO₅
411.5

VLIM-00812
10222-61-4
C₁₄H₁₅NO₃
245.27
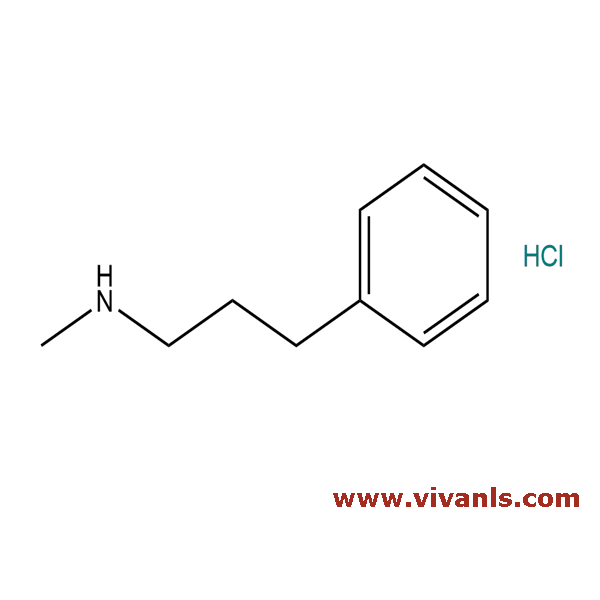
VLIM-00813
30684-07-2
C₁₀H₁₆ClN
185.69
 Isomer-1664369745.png)
VLIM-00814
1357289-37-2
C₂₀H₁₉F₂N₃O₅
419.4
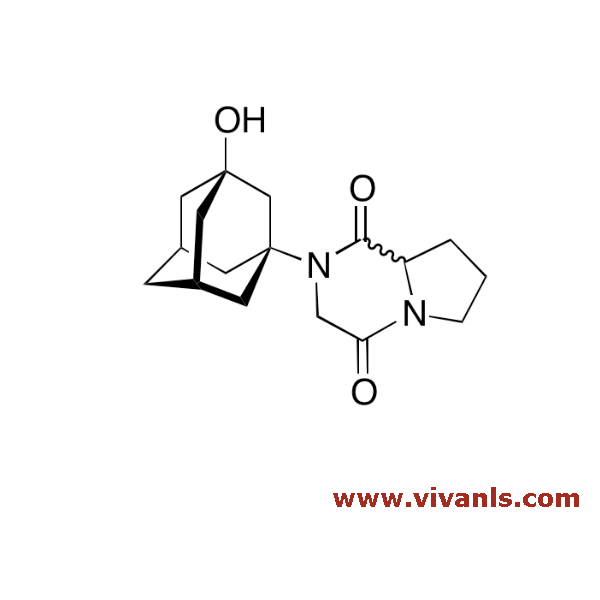
VLIM-00815
1789703-36-1
C₁₇H₂₄N₂O₃
304.38

VLIM-00816
95153-31-4
C₁₇H₂₈N₂O₅
340.41

VLIM-00817
494-52-0
C₁₀H₁₄N₂
162.23

VLIM-00818
1838139-08-4
C₁₆H₁₉N₃O₆
349.34

VLIM-00819
NA
C₂₈H₃₁N₅O₈
565.57

VLIM-00820
1838133-00-8
C₂₃H₂₁Cl₂N₃O₅S₂
554.47

VLIM-00821
1838139-24-4
C₁₉H₁₈ClN₃O₅S
435.88