VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00785
1356837-89-2
C₂₀H₃₄N₂O₅
382.5

VLIM-00786
1950-69-2
C₇H₇BrO₂S
235.10

VLIM-00787
116883-62-6
C₁₄H₁₃IO₃
356.15

VLIM-00788
270-75-7
C₈H₆O
118.13
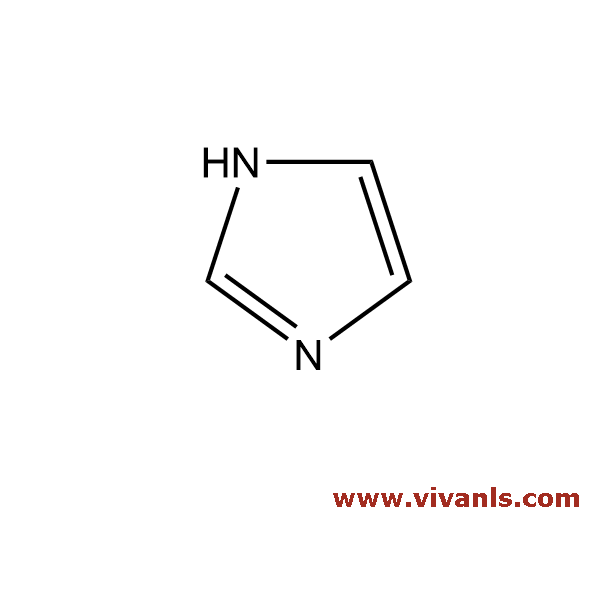

VLIM-00790
99-33-2
C₇H₃ClN₂O₅
230.56
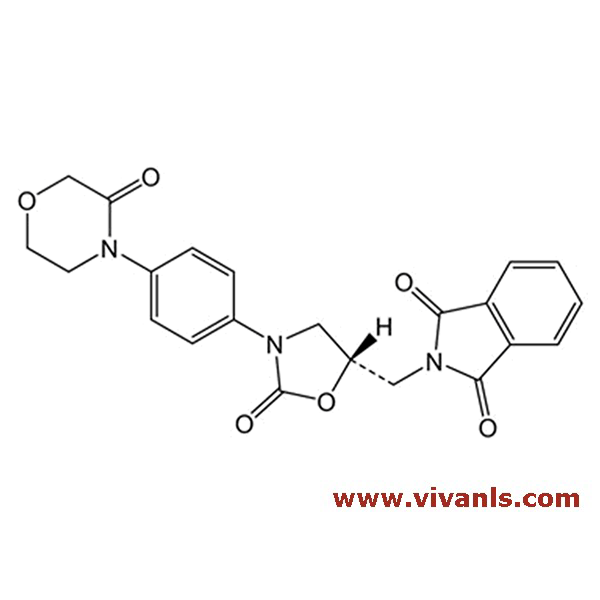
VLIM-00791
446292-08-6
C₁₈H₂₀ClN₃O₄S
409.9

VLIM-00792
1632463-24-1
C₃₈H₃₆Cl₂N₆O₁₀S₂
871.8

VLIM-00793
1151893-81-0
C₂₄H₂₁Cl₂N₃O₇S₂
598.5

VLIM-00794
1807455-76-0
C₂₅H₂₄Cl₂N₄O₆S₂
611.5

VLIM-00795
105102-22-5
C₂₂H₂₈Cl₂O₄
427.4

VLIM-00796
83881-09-8
C₂₇H₂₉ClO₆
485.0