VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
-1664255922.png)
VLIM-00749
5343-52-2
C₈H₁₆O₂
144.2

VLIM-00750
106308-41-2
C₁₀H₉FN₄O
220.2
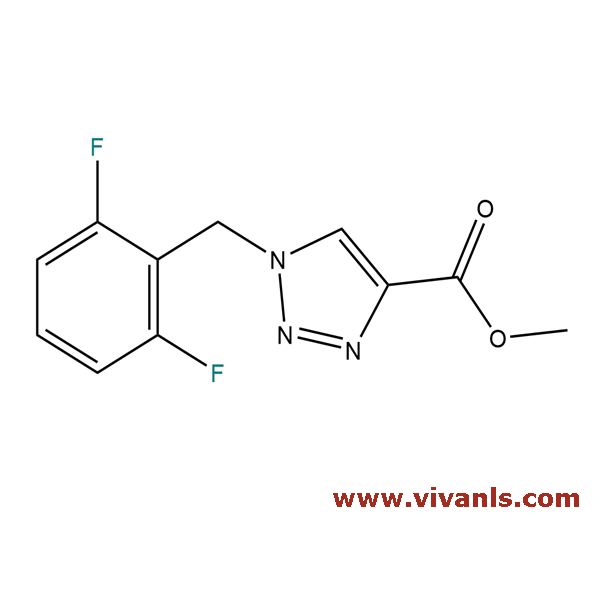
VLIM-00751
217448-86-7
C₁₁H₉F₂N₃O₂
253.2

VLIM-00752
55290-68-1
C₁₉H₂₂ClN₅O₂
387.86

VLIM-00753
62337-66-0
C₁₉H₂₃N₅O
337.42

VLIM-00754
2512213-25-9
C₁₇H₂₇ClN₂O
310.86

VLIM-00755
6323-09-7
C₂₃H₃₀Cl₂N₄
433.42
-1664256048.png)
VLIM-00756
53-86-1
C₁₉H₁₆ClNO₄
357.79
-1664256065.png)
VLIM-00757
74-11-3
C₇H₅ClO₂
156.57

VLIM-00758
155172-12-6
C₁₇H₁₉N₃O
194.23

VLIM-00759
61337-89-1
C₁₇H₂₁N₃O
283.37
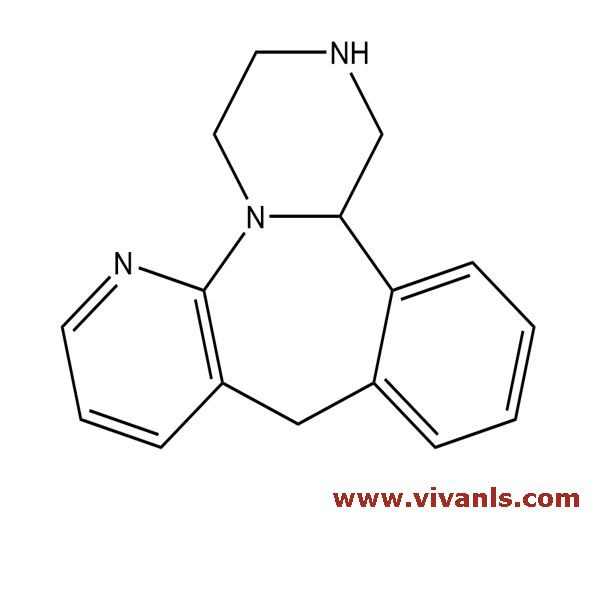
VLIM-00760
61337-68-6
C₁₆H₁₇N₃
251.33