VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00761
191546-94-8
C₁₇H₂₁N₃
267.4

VLIM-00762
191546-97-1
C₁₇H₁₇N₃O
279.34
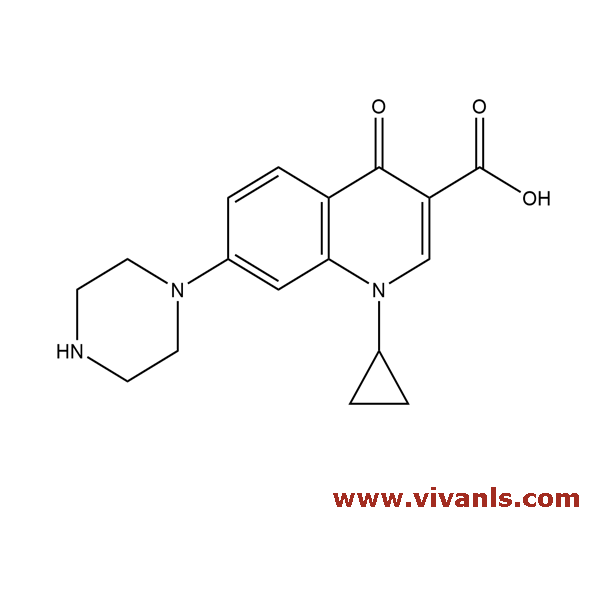
VLIM-00763
93107-11-0
C₁₇H₁₉N₃O₃
313.35

VLIM-00764
526204-10-4
C₁₇H₁₉Cl₂N₃O₃
384.26
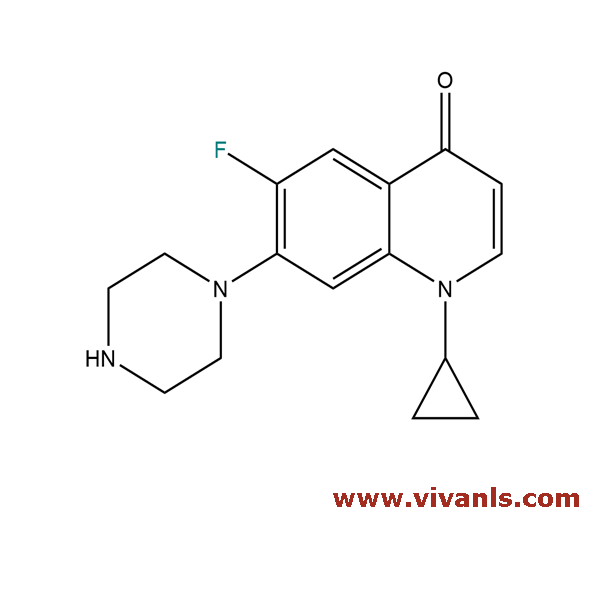
VLIM-00765
105394-83-0
C₁₆H₁₈FN₃O
287.33

VLIM-00766
226903-07-7
C₁₇H₁₉N₃O₄
329.35

VLIM-00767
90-64-2
C₈H₈O₃
152.15
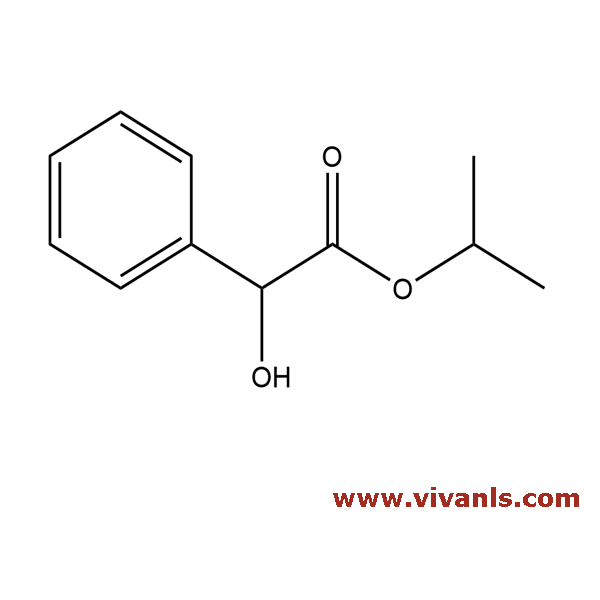
VLIM-00768
4118-51-8
C₁₁H₁₄O₃
194.23
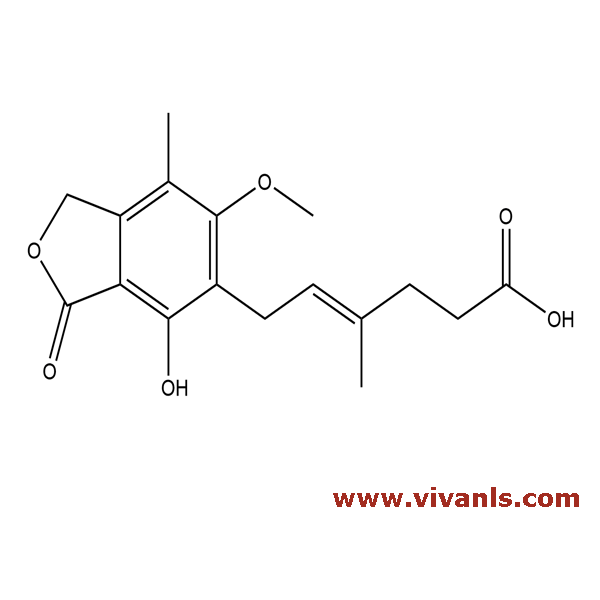
VLIM-00769
24280-93-1
C₁₇H₂₀O₆
320.34

VLIM-00770
2471-70-7
C₁₂H₁₀O₃
202.21
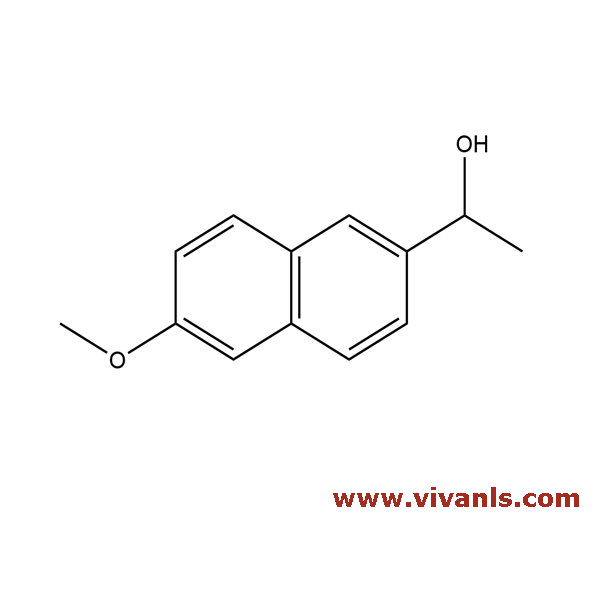
VLIM-00771
77301-42-9
C₁₃H₁₄O₂
202.25

VLIM-00772
3900-45-6
C₁₃H₁₂O₂
200.23