VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00737
168828-89-5
C₂₂H₂₀FN₃O₅
425.41

VLIM-00738
168828-90-8
C₁₄H₁₈FN₃O₃
295.31

VLIM-00739
1271930-15-4
C₃₂H₃₁F₆N
543.6

VLIM-00740
69224-79-9
C₂₅H₃₃ClO₆
464.98
-1664255576.png)
VLIM-00741
6901-13-9
C₂₂H₂₅NO₆
399.44

VLIM-00742
120503-69-7
C₈H₁₀N₆
190.21
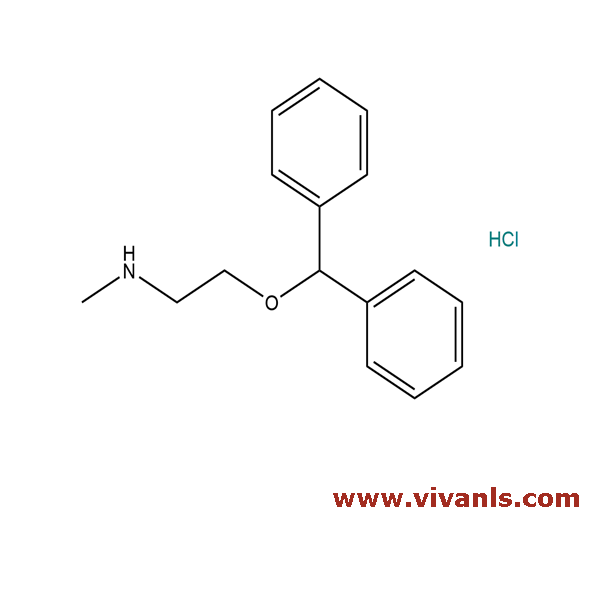
VLIM-00743
53499-40-4
C₁₆H₂₀ClNO
277.79
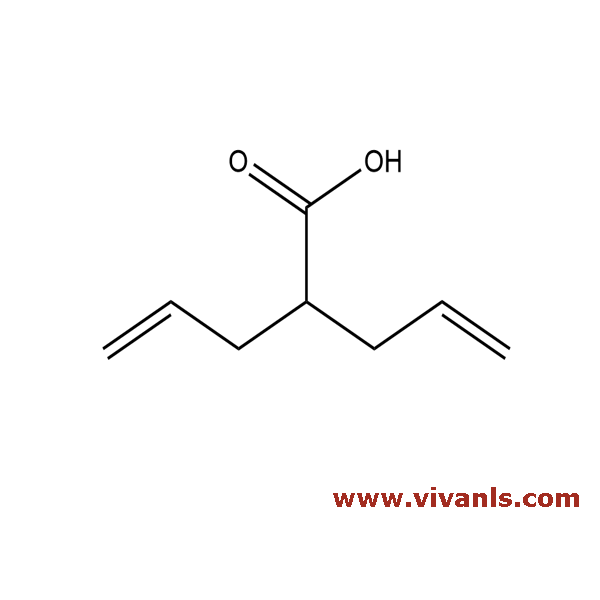
VLIM-00744
99-67-2
C₈H₁₂O₂
'140.2

VLIM-00745
107-92-6
C₄H₈O₂
88.11

VLIM-00746
5534-18-9
C₂₅H₃₃ClO₆
464.98

VLIM-00747
110104-37-5
C₁₀H₁₃N₅O₄
267.25

VLIM-00748
99462-32-5
C₁₂H₉N₃O₂
227.22