VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00701
876473-73-3
C₁₂H₁₈O
178.27

VLIM-00702
94240-17-2
C₉H₁₂ClNO₂
'201.65

VLIM-00703
71786-67-9
C₁₆H₁₈ClNO₂
291.77

VLIM-00704
'1916-07-0
C₁₁H₁₄O₅
'226.23

VLIM-00705
616-47-7
C₄H₆N₂
82.1
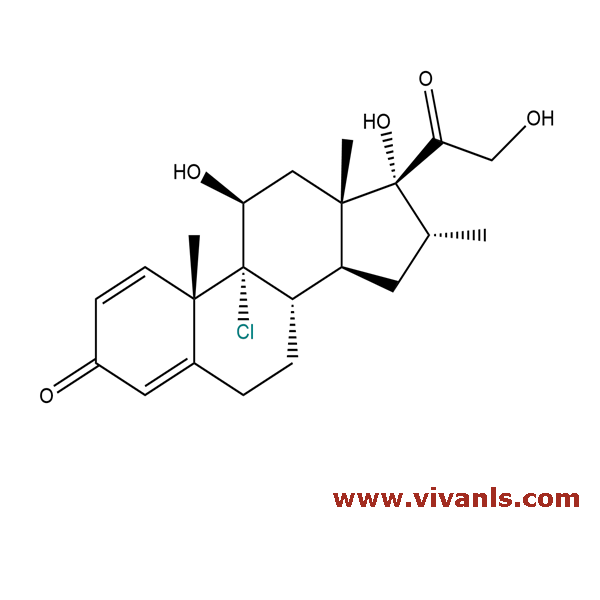
VLIM-00706
4647-20-5
C₂₂H₂₉ClO₅
408.92

VLIM-00707
83881-08-7
C₂₂H₂₇ClO₄
390.9
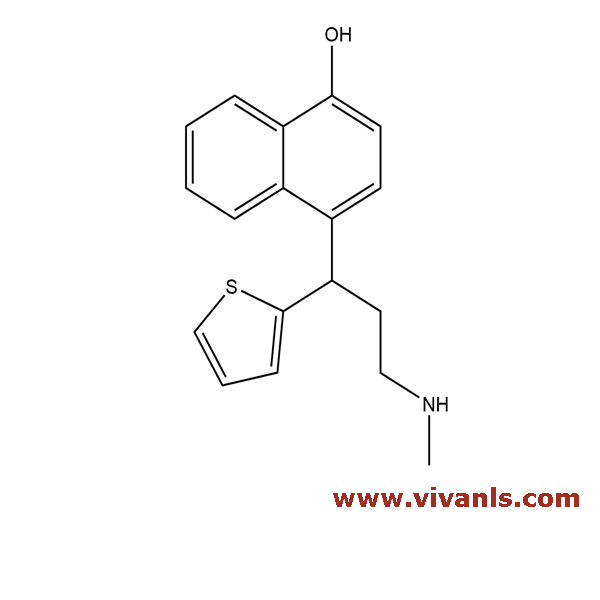
VLIM-00708
949095-98-1
C₁₈H₁₉NOS
297.41

VLIM-00709
959392-22-4
C₁₈H₁₉NOS
297.41

VLIM-00710
18910-68-4
C₁₃H₂₁NO₂
223.32

VLIM-00711
24085-03-8
C₂₀H₂₇NO₃
329.43
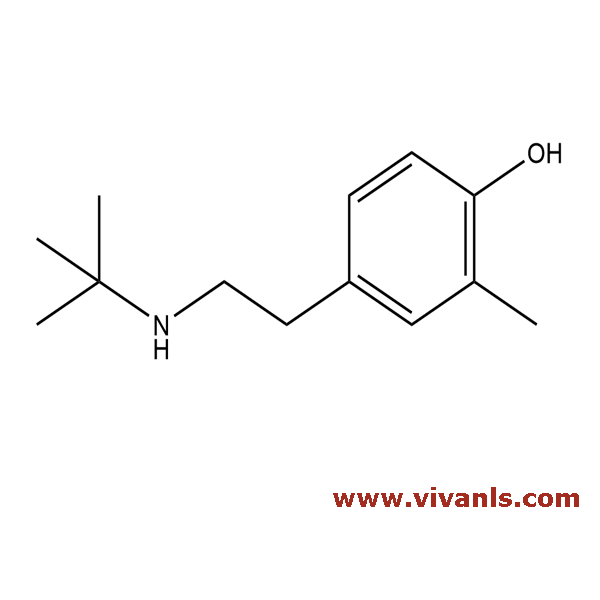
VLIM-00712
132183-64-3
C₁₃H₂₁NO
207.31