VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
-Isomer-1664196694.png)
VLIM-00689
1593542-96-1
C₂₄H₂₁F₂NO₃
409.43

VLIM-00690
163380-16-3
C₂₄H₂₁F₂NO₃
409.43
-Isomer-1664196751.png)
VLIM-00691
1376614-99-1
C₂₄H₂₁F₂NO₃
409.43

VLIM-00692
1593543-00-0
C₂₄H₂₁F₂NO₃
409.43

VLIM-00693
1370335-38-8
C₂₂H₂₀Cl₄N₂O₃
502.21

VLIM-00694
67358-54-7
C₁₅H₁₄Cl₅NO
401.53
-1664196853.png)
VLIM-00695
934365-23-8
C₃₆H₄₂Cl₂N₂
573.64
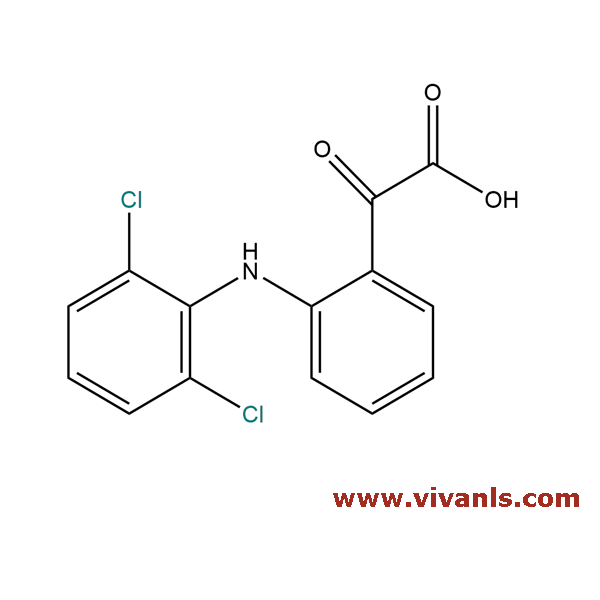
VLIM-00696
66156-75-0
C₁₄H₉Cl₂NO₃
310.13

VLIM-00697
59804-37-4
C₁₃H₁₁N₃O₄S₂
337.37

VLIM-00698
120279-90-5
C₁₀H₁₆N₂O₄S₃
324.44

VLIM-00699
612069-25-7
C₃₆H₆₅NO₁₃
719.9

VLIM-00700
108-32-7
C₄H₆O₃
'102.09