VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
-1664196238.png)
VLIM-00665
'2125704-30-3
C₁₄H₉ClF₃NO₂
315.7

VLIM-00666
353270-76-5
C₁₅H₁₁ClF₃NO₂
329.7

VLIM-00667
109632-08-8
C₁₄H₂₃NO₃
253.34
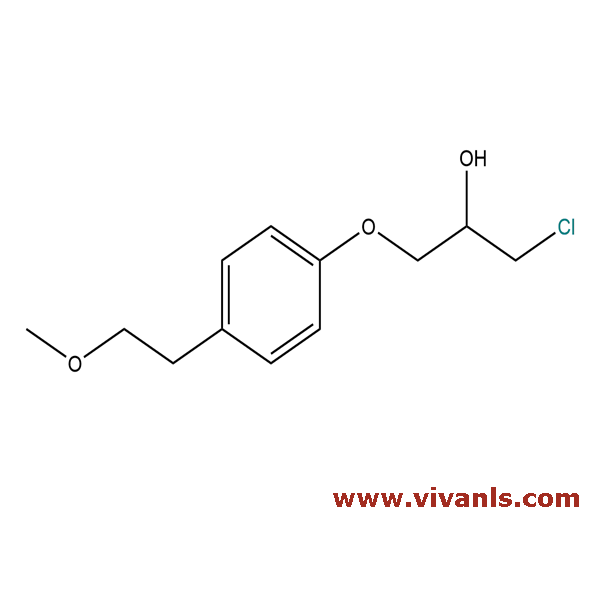
VLIM-00668
56718-76-4
C₁₂H₁₇ClO₃
244.71

VLIM-00669
29122-74-5
C₁₃H₁₉NO₃
237.29

VLIM-00670
1486464-40-7
C₂₇H₄₁NO₆ • HCl
475.62

VLIM-00671
106685-41-0
C₂₉H₃₀O₃
426.55

VLIM-00672
932033-58-4
C₂₉H₃₀O₃
426.55

VLIM-00673
1453-82-3
C₆H₆N₂O
122.12

VLIM-00674
462-08-8
C₅H₆N₂
94.11

VLIM-00675
121-00-6
C₁₁H₁₆O₂
180.24

VLIM-00676
121-71-1
C₈H₈O₂
136.15