VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
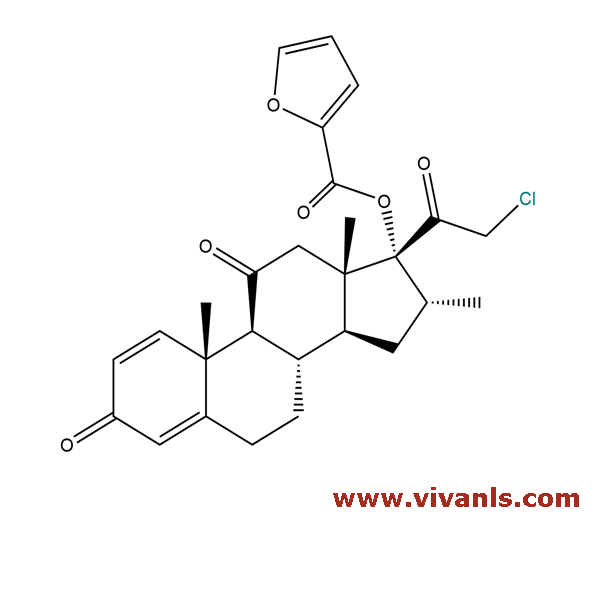
VLIM-00641
1305334-31-9
C₂₇H₂₉ClO₆
484.97
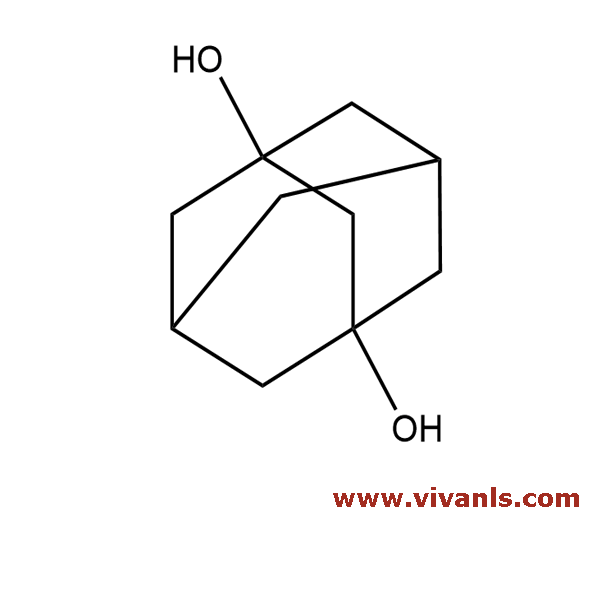
VLIM-00642
5001-18-3
C₁₀H₁₆O₂
168.23
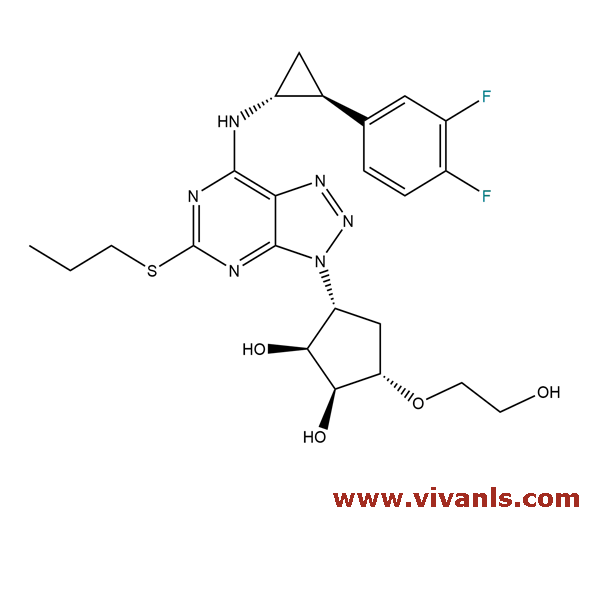
VLIM-00643
274693-27-5
C₂₃H₂₈F₂N₆O₄S
522.57

VLIM-00644
274693-26-4
C₂₆H₃₂F₂N₆O₄S
562.63

VLIM-00645
1788033-05-5
C₂₃H₂₈F₂N₆O₄S
522.57

VLIM-00646
754926-25-5
C₁₅H₂₅NO₃
267.36

VLIM-00647
191939-34-1
C₁₆H₂₀ClN₃O₂S.HCl
390.33

VLIM-00648
65322-85-2
C₁₃H₁₈O₂
206.29

VLIM-00649
1803026-58-5
C₁₆H₁₂F₆N₄O
390.28
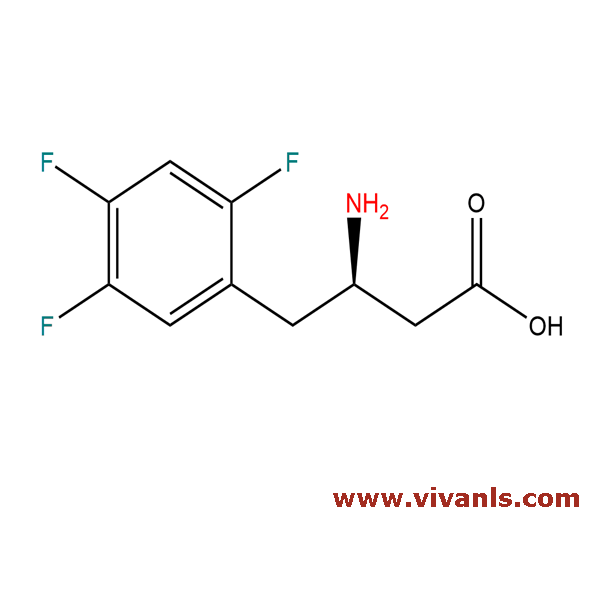
VLIM-00650
936630-57-8
C₁₀H₁₀F₃NO₂
233.19

VLIM-00651
2088771-60-0
C₂₀H₁₉F₆N₅O₅
523.39

VLIM-00652
NA
C₂₅H₂₇N₇O₂
457.53