VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00677
565453-39-6
C₁₇H₂₇N₃O₃
321.41
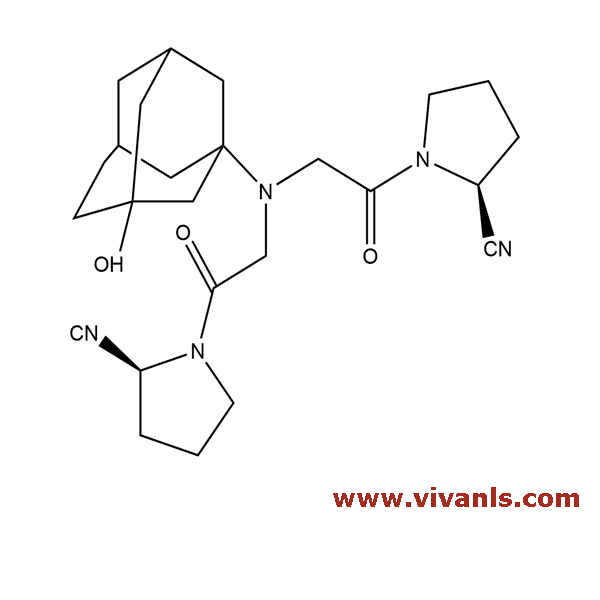
VLIM-00678
1036959-23-5
C₂₄H₃₃N₅O₃
439.55
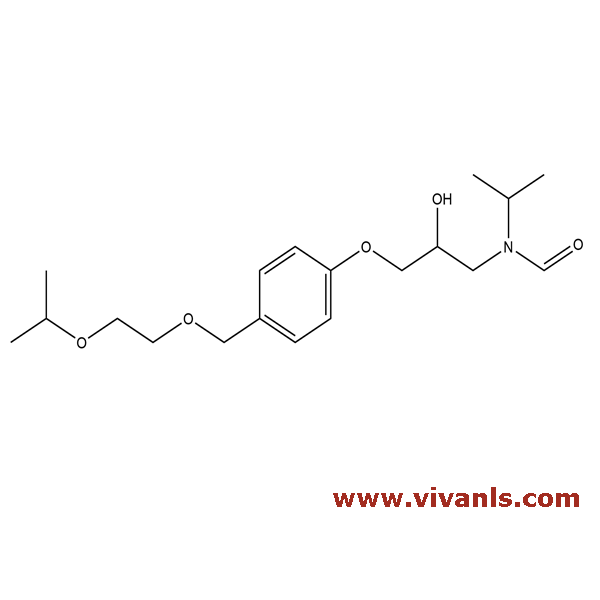
VLIM-00679
1447715-45-8
C₁₉H₃₁NO₅
353.45
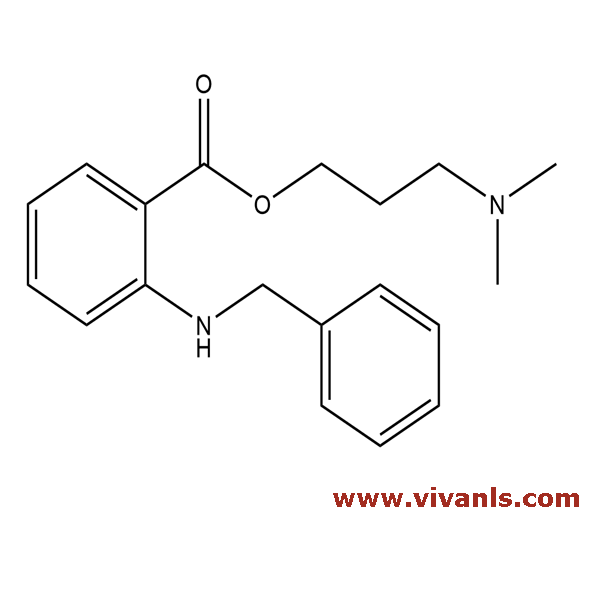
VLIM-00680
87453-76-7
C₁₉H₂₄N₂O₂
312.41

VLIM-00681
1337966-15-0
C₂₃H₃₂N₄O
'380.50

VLIM-00682
87453-75-6
C₁₂H₁₈N₂O₂
222.14

VLIM-00683
'1391054-37-7
C₁₁H₉FO₂
192.19

VLIM-00684
1391052-75-7
C₂₀H₂₁ClFNO₃S
373.44

VLIM-00685
302781-98-2
C₂₄H₂₁F₂NO₃
409.43

VLIM-00686
1798008-25-9
C₂₄H₂₁F₂NO₃
409.43

VLIM-00687
1700622-06-5
C₂₄H₂₁F₂NO₃
409.43
-Isomer-1664196679.png)
VLIM-00688
1593543-07-7
C₂₄H₂₁F₂NO₃
409.43