VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00713
684286-46-2
C₂₄H₃₄N₄O₅S
490.63

VLIM-00714
57969-05-8
C₁₈H₂₃NO₂
285.38

VLIM-00715
24155-42-8
C₁₁H₁₀Cl₂N₂O
257.12

VLIM-00716
22916-47-8
C₁₈H₁₄Cl₄N₂O
416.13

VLIM-00717
18910-70-8
C₁₄H₂₃NO₃
253.34

VLIM-00718
56796-66-8
C₂₀H₂₇NO₃
329.43
 EP impurity J-1664197320.png)
VLIM-00719
1331637-48-9
C₁₁H₂₂N₆O₄S₂
366.46

VLIM-00720
1305320-62-0
C₁₅H₁₃Cl₄NO
365.08
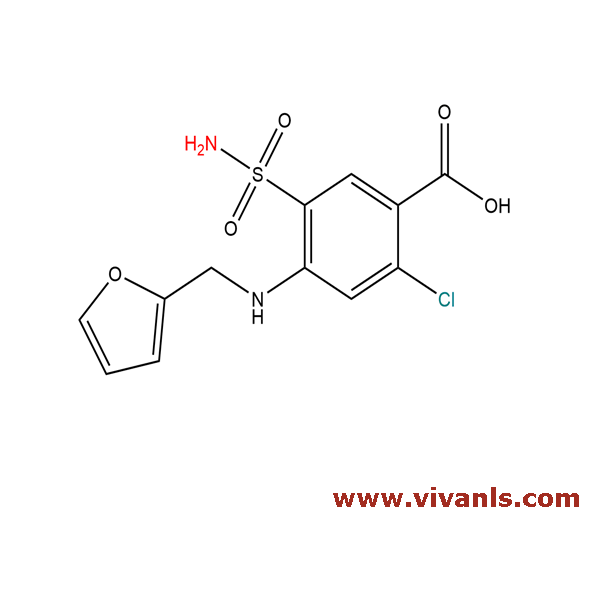
VLIM-00721
4818-59-1
C₁₂H₁₁ClN₂O₅S
330.74

VLIM-00722
2736-23-4
C₇H₅Cl₂NO₄S
270.09

VLIM-00723
3086-91-7
C₇H₇ClN₂O₄S
250.66

VLIM-00724
5046-19-5
C₁₇H₁₇N₃O₆S
391.4