VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00616
131918-64-4
C₂₅H₃₂O₆
428.52
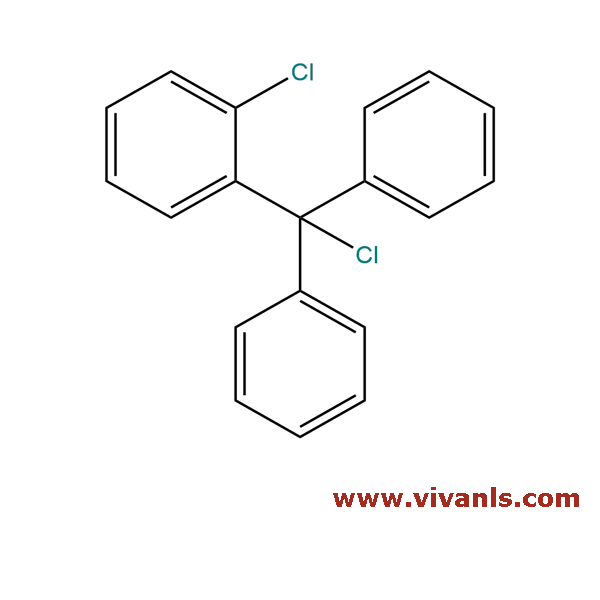
VLIM-00617
42074-68-0
C₁₉H₁₄Cl₂
313.22
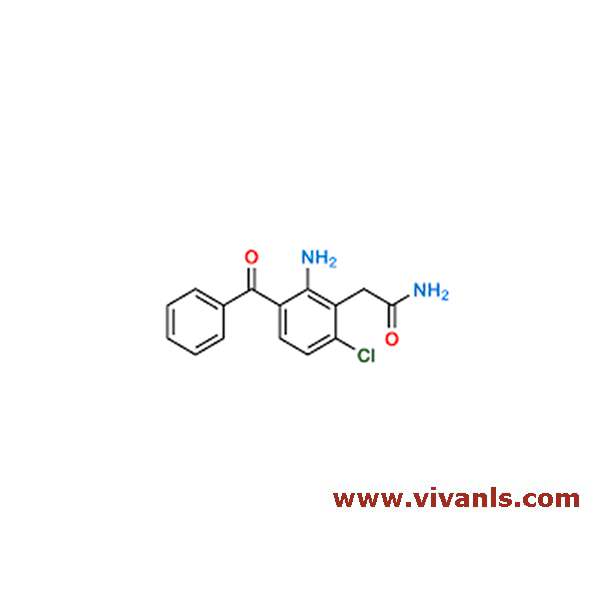
VLIM-00618
2731586-58-4
C₁₅H₁₃ClN₂O₂
288.73

VLIM-00619
50892-62-1
C₁₃H₉ClN₂O
244.7

VLIM-00620
263366-81-0
C₃₀H₂₄Cl₂N₆
'539.50
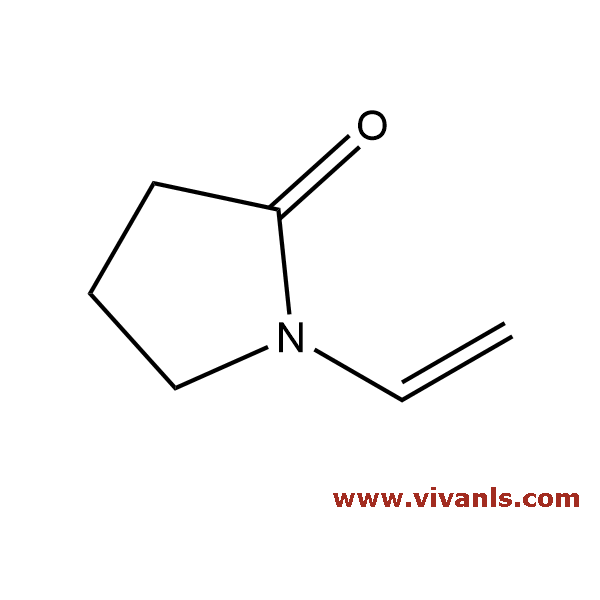
VLIM-00621
'88-12-0
C₆H₉NO
111.14

VLIM-00622
'504-29-0
C₅H₆N₂
'C5H6N2

VLIM-00623
6298-37-9
C₈H₇N₃
145.16

VLIM-00624
842138-74-3
C₉H₇BrN₄S
283.15

VLIM-00625
65514-71-8
C₁₈H₂₁ClN₄O
344.9

VLIM-00626
'1977-07-7
C₁₈H₂₀N₄
292.38

VLIM-00627
299409-85-1
C₃₇H₆₇NO₁₃
733.93