VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00592
106649-96-1
C₆H₁₂Cl₂N₆S₂
303.24
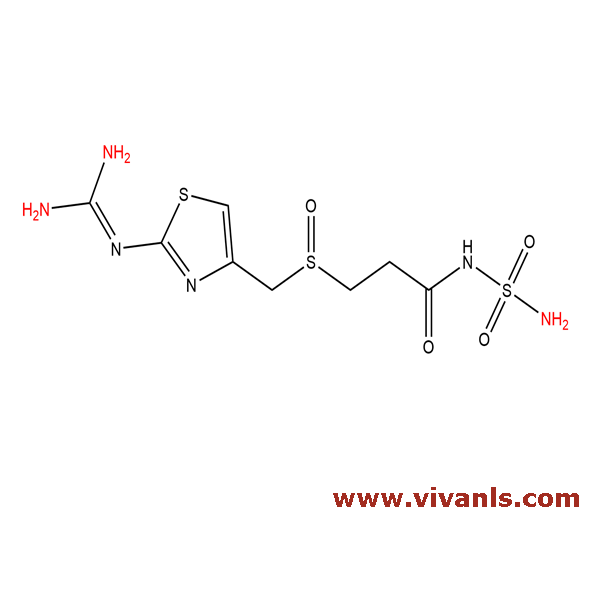
VLIM-00593
1020719-36-1
C₈H₁₄N₆O₄S₃
354.43

VLIM-00594
122111-05-1
C₉H₁₂ClF₂N₃O₄
299.66

VLIM-00595
'945553-91-3
C₂₄H₂₅ClFN₅O₃
485.94

VLIM-00596
129722-12-9
C₂₃H₂₅Cl₂N₃O₂
'446.37

VLIM-00597
573691-09-5
C₂₃H₂₇Cl₂N₃O₃
464.38

VLIM-00598
696-23-1
C₄H₅N₃O₂
'127.10

VLIM-00599
5006-69-9
C₅H₇N₃O₃
157.13

VLIM-00600
5006-68-8
C₅H₇N₃O₃
157.13
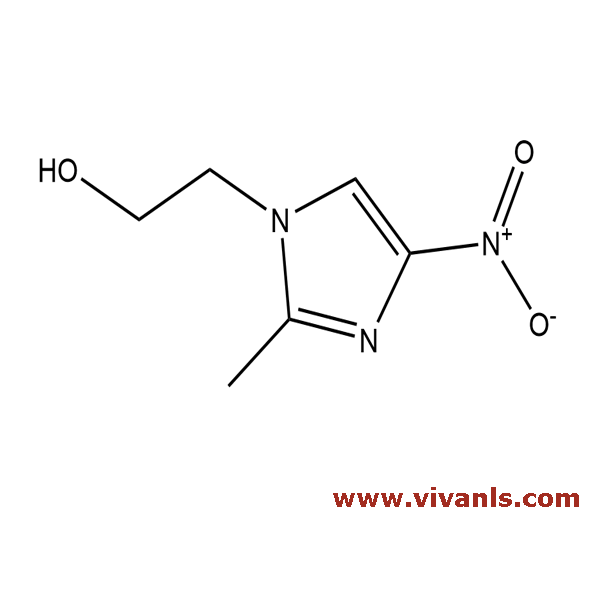
VLIM-00601
705-19-1
C₆H₉N₃O₃
171.15
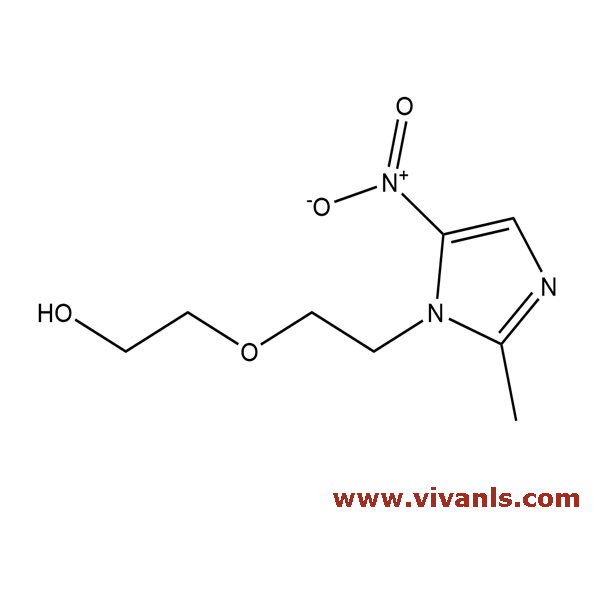
VLIM-00602
16156-94-8
C₈H₁₃N₃O₄
215.21

VLIM-00603
1010-93-1
C₆H₇N₃O₄
185.14