VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
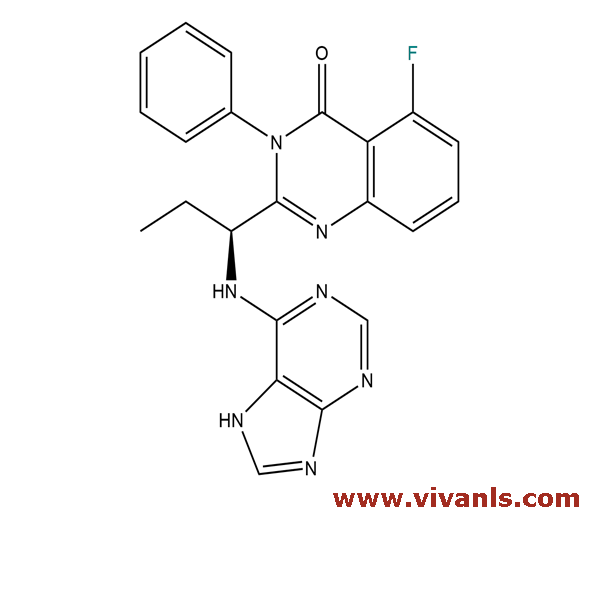
VLIM-00604
2184933-01-3
C₂₂H₁₈FN₇O
415.42

VLIM-00605
202409-31-2
C₁₇H₁₃ClN₂O₂S
'344.82

VLIM-00606
221615-75-4
C₁₅H₁₅NO₃S
289.35

VLIM-00607
52079-10-4
C₁₃H₁₂O₃
216.23

VLIM-00608
NA
C₂₂H₂₃FN₂O₅
'414.43

VLIM-00609
66774-02-5
C₁₉H₁₅ClO
294.77

VLIM-00610
23593-71-7
C₂₂H₁₇ClN₂
344.84

VLIM-00611
1616703-93-5
C₂₅H₃₀F₂N₆O₅S
564.61
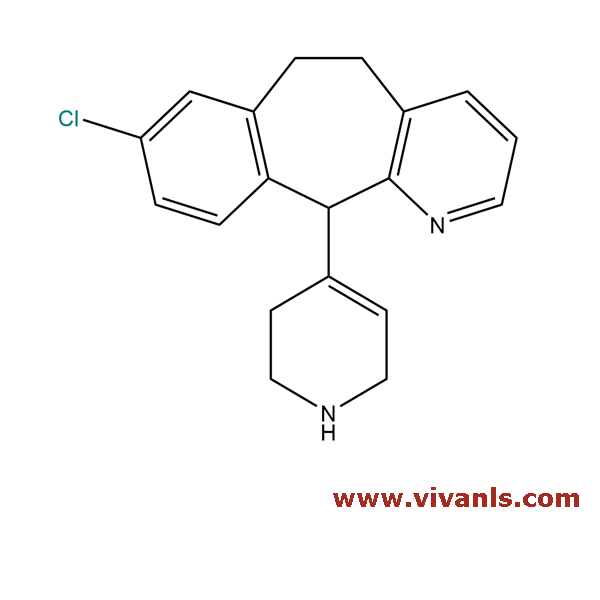
VLIM-00612
432543-89-0
C₁₉H₂₀Cl₂N₂
346.58

VLIM-00613
NA
C₂₁H₁₅NO₅
361.34

VLIM-00614
118457-16-2
C₂₂H₂₅F₂NO₄
405.44

VLIM-00615
145195-63-7
C₁₄H₁₅N₃O₅
305.29