VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00628
84-66-2
C₁₂H₁₄O₄
222.24
-4-Bromomethylbiphenyl– Olmesartan Impurity-1664195590.png)
VLIM-00630
88546-55-8
C₁₇H₁₉N₃O₄S
361.42
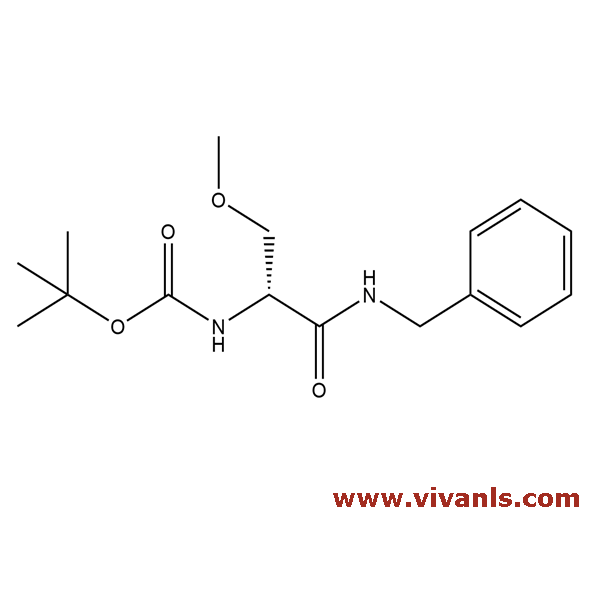

VLIM-00632
NA
C₁₄H₂₀N₂O₃
264.33

VLIM-00633
30536-19-7
C₆H₄ClN₃S
185.63

VLIM-00634
51323-05-8
C₇H₅ClN₄S₂
244.72

VLIM-00635
51322-69-1
C₉H₉N₅S
219.27

VLIM-00636
51323-03-6
C₈H₈ClIN₄S₂
386.66

VLIM-00637
107-15-3
C₂H₉ClN₂
156.66

VLIM-00638
1500558-49-5
C₁₀H₁₉NO₂.HCl
221.72

VLIM-00639
51322-80-6
C₇H₃ClN₄S
210.64

VLIM-00640
NA
C₂₆H₃₇ClN₂O₁₀ • C₂H₄O₂
633.08