VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

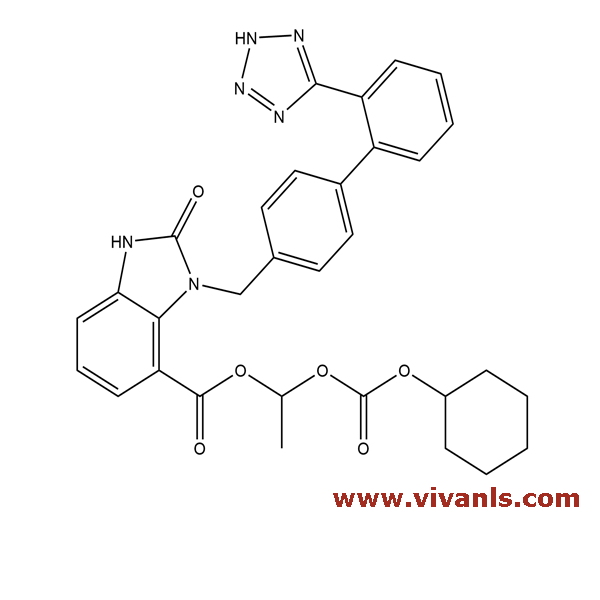
VLIM-00568
139481-58-6
C₂₆H₂₄N₆O₃
468.51
VLIM-00569
869631-11-8
C₃₁H₃₀N₆O₆
'582.61

VLIM-00570
1185255-99-5
C₃₃H₃₄N₆O₆
'610.66

VLIM-00571
1185256-03-4
C₃₃H₃₄N₆O₆
610.66

VLIM-00572
914613-35-7
C₃₅H₃₈N₆O₆
638.71

VLIM-00573
914613-36-8
C₃₅H₃₈N₆O₆
638.71

VLIM-00574
139481-59-7
C₂₄H₂₀N₆O₃
'440.45

VLIM-00575
170791-09-0
C₅₂H₄₈N₆O₆
852.97

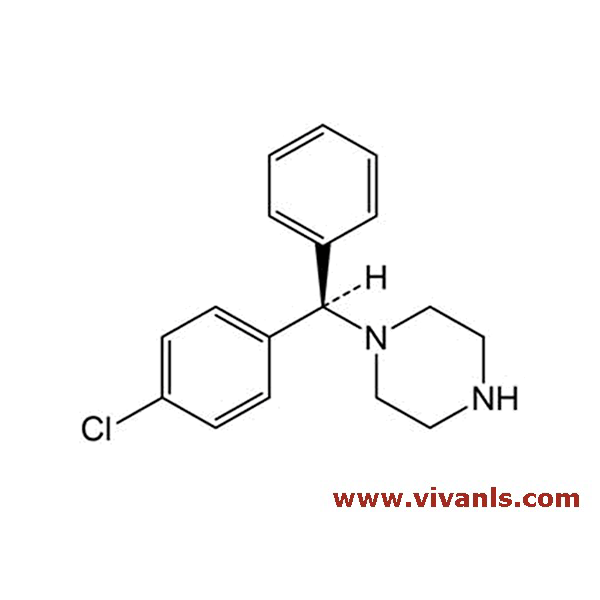
VLIM-00576
38092-89-6
C₂₀H₂₁ClN₂
324.85
VLIM-00577
300543-56-0
C₁₇H₁₉ClN₂
286.8

VLIM-00578
942193-17-1
C₁₉H₂₁ClN₂O₂
344.84

VLIM-00579
NA
C₂₁H₂₅ClN₂O₃
388.9