VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00531
16339-04-1
C₅H₁₂N₂O
116.16

VLIM-00532
61445-55-4
C₅H₁₀N₂O₃
146.15

VLIM-00533
100-75-4
C₅H₁₀N₂O
114.15

VLIM-00534
924-16-3
C₈H₁₈N₂O
158.24
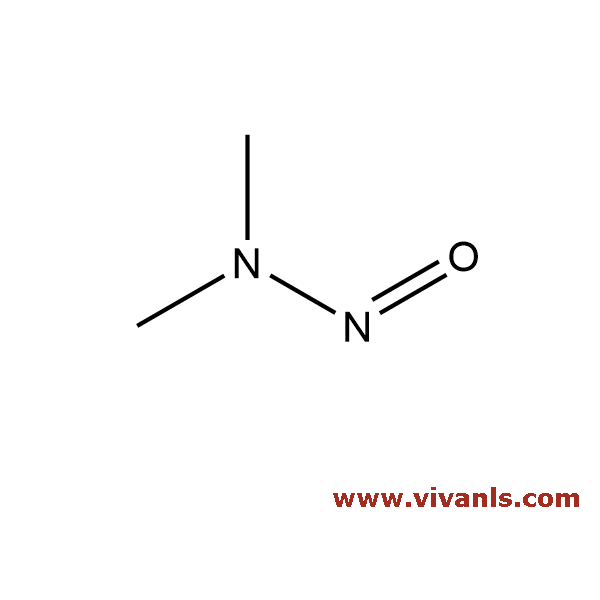
VLIM-00535
62-75-9
C₂H₆N₂O
74.08

VLIM-00537
100-75-4
C₅H₁₀N₂O
114.15

VLIM-00538
621-64-7
C₆H₁₄N₂O
'130.19

VLIM-00539
NA
C₂₄H₃₃N₅O₃
439.55

VLIM-00540
1251765-07-7
C₁₄H₂₂N₆O₄S
370.43
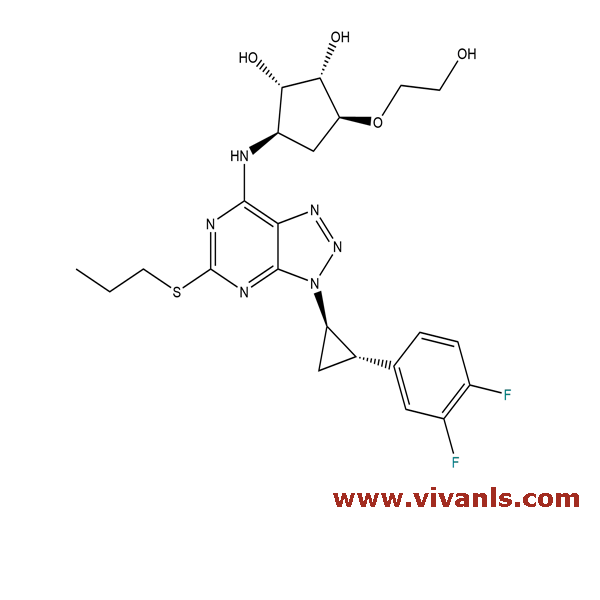
VLIM-00541
1788033-05-5
C₂₃H₂₈F₂N₆O₄S
522.57
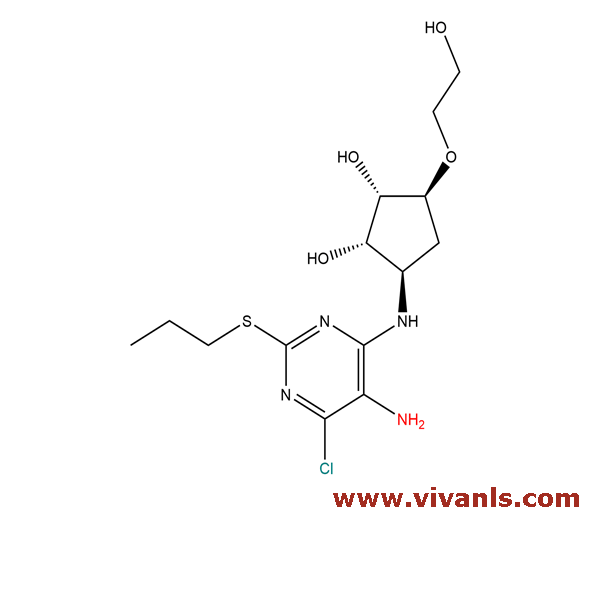
VLIM-00542
1402150-32-6
C₁₄H₂₃ClN₄O₄S
'378.87
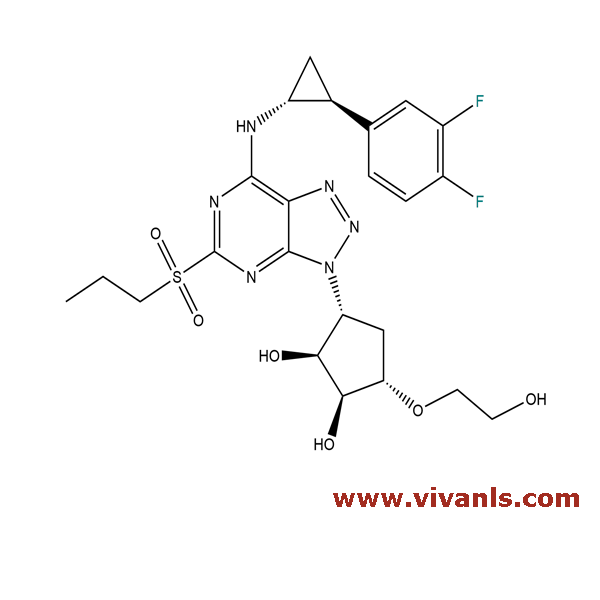
VLIM-00543
274693-39-9
C₂₃H₂₈F₂N₆O₆S
554.57