VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00446
'763924-54-5
C₃₉H₇₄N₂O₁₂
763.01

VLIM-00447
NA
C₂₉H₄₀N₂O₄
'382.14

VLIM-00448
170570-01-1
C₁₇H₁₄F₃N₃O₂S
381.37

VLIM-00449
16899-81-3
C₁₁H₁₅NO₃.HCl
'245.70
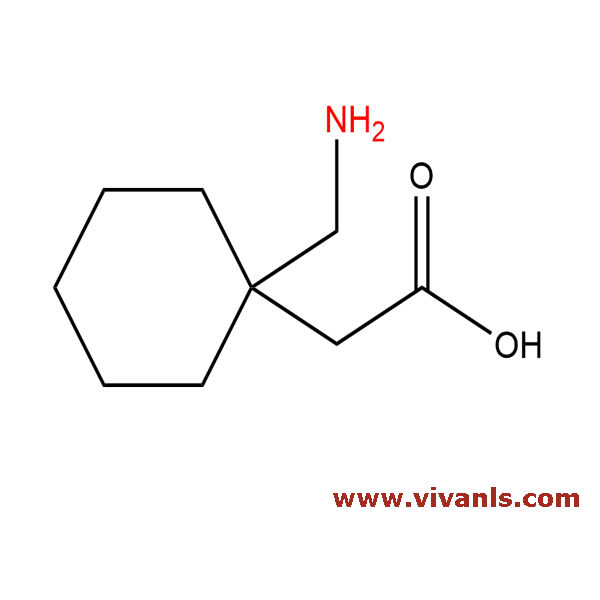
VLIM-00450
60142-96-3
C₉H₁₇NO₂
'171.24

VLIM-00451
1500558-49-5
C₁₀H₁₉NO₂.HCl
221.72

VLIM-00452
3506-32-9
C₁₁H₁₇NO₂
195.26
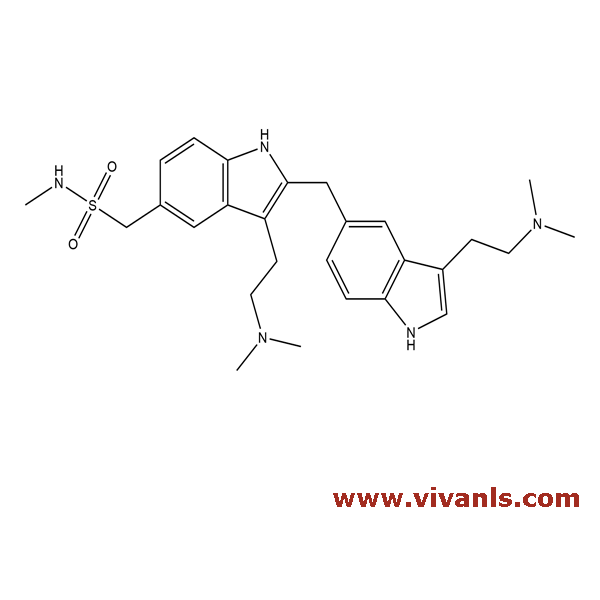
VLIM-00453
545338-89-4
C₂₇H₃₇N₅O₂S
495.68

VLIM-00454
88919-51-1
C₁₃H₁₉N₃O₂S
281.37

VLIM-00455
1797905-62-4
C₁₅H₂₃N₃O₃S
325.43
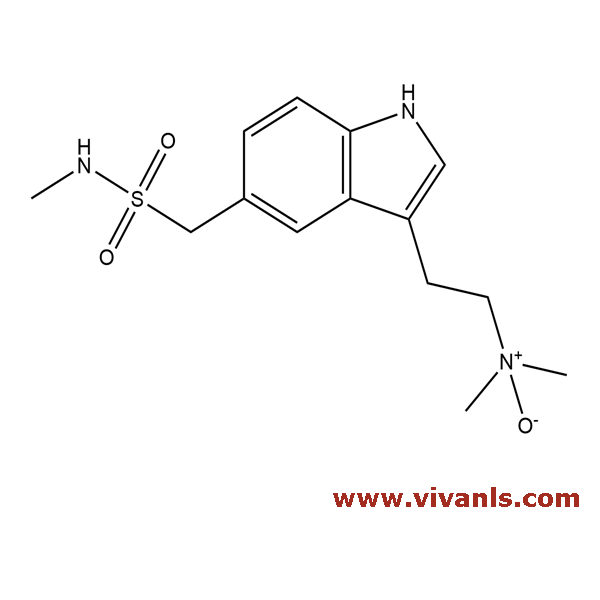
VLIM-00456
212069-94-8
C₁₄H₂₁N₃O₃S
311.4

VLIM-00457
88919-22-6
C₁₂H₁₇N₃O₂S
267.35