VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00422
54060-30-9
C₈H₇N
117.15

VLIM-00423
152460-10-1
C₁₆H₁₅N₅
'277.32

VLIM-00424
581076-65-5
C₂₁H₂₈N₆O
'380.49

VLIM-00425
199327-61-2
C₁₆H₂₁N₃O₄
319.36

VLIM-00426
1603814-04-5
C₂₂H₂₄ClFN₄O₃
446.9

VLIM-00427
42399-49-5
C₁₆H₁₅NO₃S
301.36

VLIM-00428
872728-81-9
C₃₄H₄₁N₇O₅ • CH ₄O₃S
627.75 + 96.10

VLIM-00429
NA
C₂₇H₂₉N₇O₃
499.56

VLIM-00430
211915-84-3
C₂₇H₂₆N₆O₃
482.53

VLIM-00431
1381757-44-3
C₃₄H₄₁N₇O₆
643.73
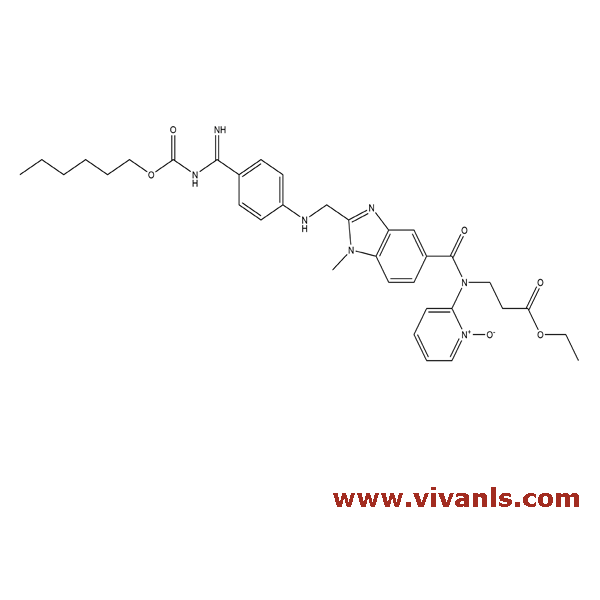
VLIM-00432
14366-59-7
C₁₂H₁₈O
178.27

VLIM-00433
201166-22-5
C₁₂H₁₆O₂
192.25