VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
![Impurities-Pramipexole EP Impurity C [Pramipexole Dimer Impurity]-1664192848.png](/images/productimages/Impurities-Pramipexole EP Impurity C [Pramipexole Dimer Impurity]-1664192848.png)
VLIM-00458
1973461-14-1
C₂₀H₃₂N₆S₂
420.64

VLIM-00459
4562-36-1
C₄₁H₆₄O₁₄
780.94

VLIM-00462
462-08-8
C₅H₆N₂
94.11

VLIM-00464
214398-99-9
C₇H₁₁ClN₂O₂
190.63
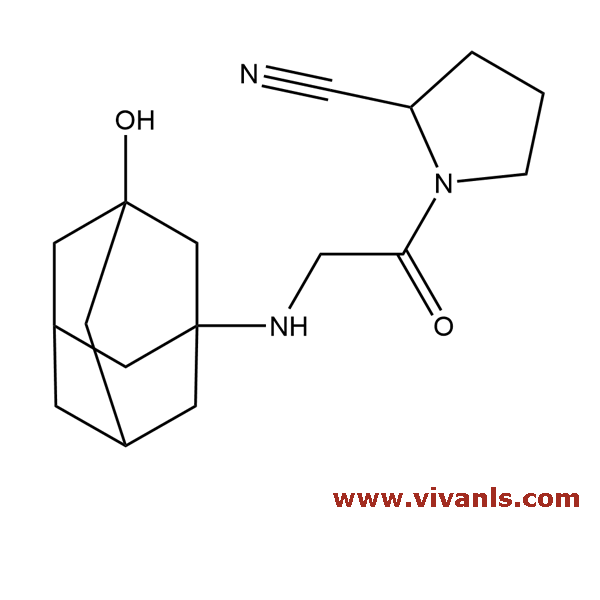
VLIM-00466
1036959-27-9
C₁₇H₂₅N₃O₂
303.41

VLIM-00467
10595-95-6
C₃H₈N₂O
88.11

VLIM-00468
55290-67-0
C₁₉H₂₄ClN₅O₃
405.88
-1664193239.png)
VLIM-00508
NA
C₁₁H₁₁FO₂
'194.02

VLIM-00524
103146-25-4
C₂₀H₃₂N₆S₂
'342.41

VLIM-00525
55-18-5
C₄H₁₀N₂O
102.13

VLIM-00527
621-64-7
C₆H₁₄N₂O
130.19

VLIM-00529
207557-35-5
C₇H₉ClN₂O
172.61