VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00410
992-62-1
C₃₆H₆₅NO₁₃
719.9

VLIM-00411
992-65-4
C₃₇H₆₇NO₁₄
749.93
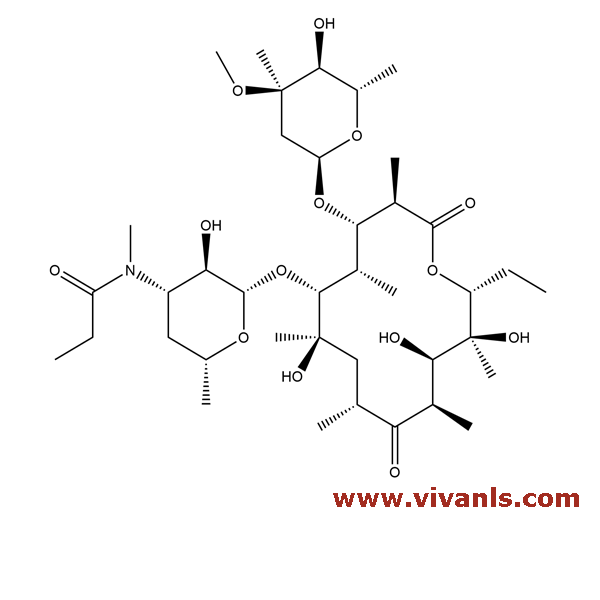
VLIM-00412
147702-49-6
C₄₂H₇₃NO₁₇
864.04

VLIM-00413
82230-93-1
C₃₇H₆₇NO₁₄
749.94
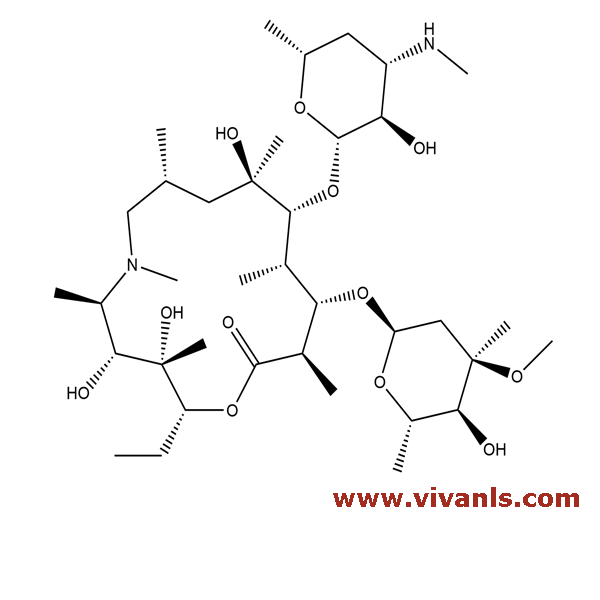
VLIM-00414
172617-84-4
C₃₇H₇₀N₂O₁₂
734.96

VLIM-00415
612069-26-8
C₃₈H₇₂N₂O₁₃
764.98

VLIM-00416
2461-42-9
C₁₃H₁₂O₂
200.23

VLIM-00417
7008-85-7
C₄H₃N₅O
137.1
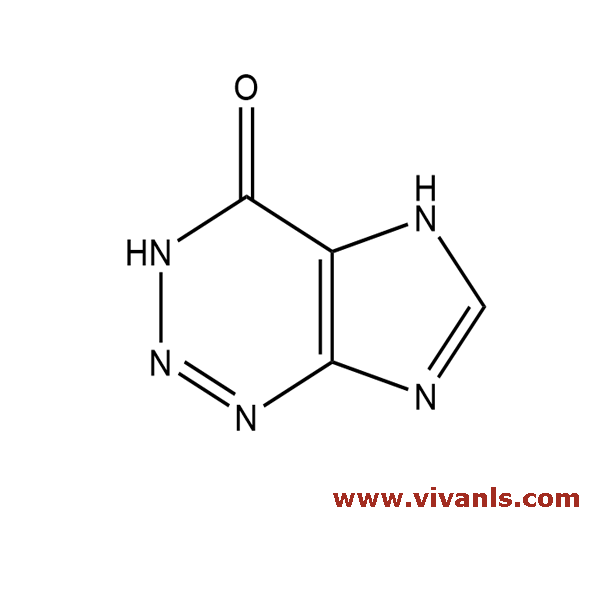
VLIM-00418
4656-86-4
C₄H₃N₅O
137.1

VLIM-00419
170727-59-0
C₂₂H₂₃ClN₂O₂
382.88

VLIM-00420
179688-29-0
C14H18N2O5
294.3

VLIM-00421
1354727-63-1
C₂₂H₂₅N₃O₅
411.46