VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
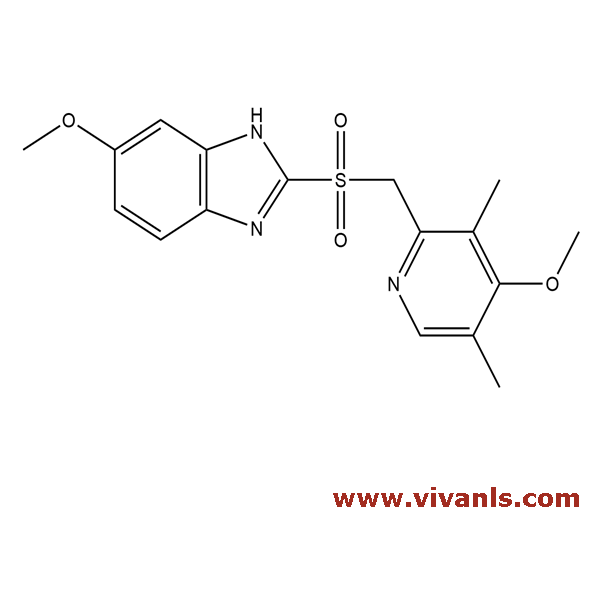
VLIM-00373
88546-55-8
C₁₇H₁₉N₃O₄S
361.42

VLIM-00374
127780-16-9
C₁₆H₁₅F₂N₃O₅S
399.37

VLIM-00375
102625-64-9
C₁₆H₁₅F₂N₃O₃S
367.37

VLIM-00376
440124-96-9
C₁₄H₁₁ClF₃NO₂
'317.69

VLIM-00377
87447-47-0
C₁₈H₁₇NO₄S
'343.40
-1664188739.png)
VLIM-00378
887196-29-4
C₆₈H₇₂F₂N₄O₁₀• Ca
1143.32 + 40.08
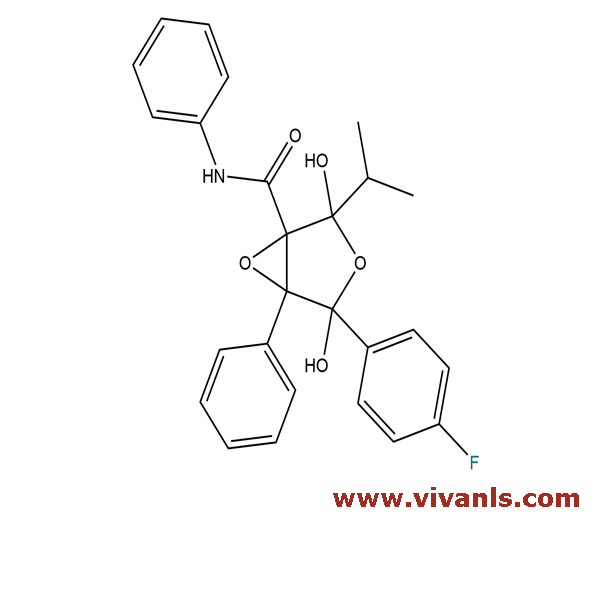
VLIM-00379
873950-19-7
C₂₆H₂₄FNO₅
449.47

VLIM-00380
NA
C₃₂H₃₈N₄O₄S
574.73
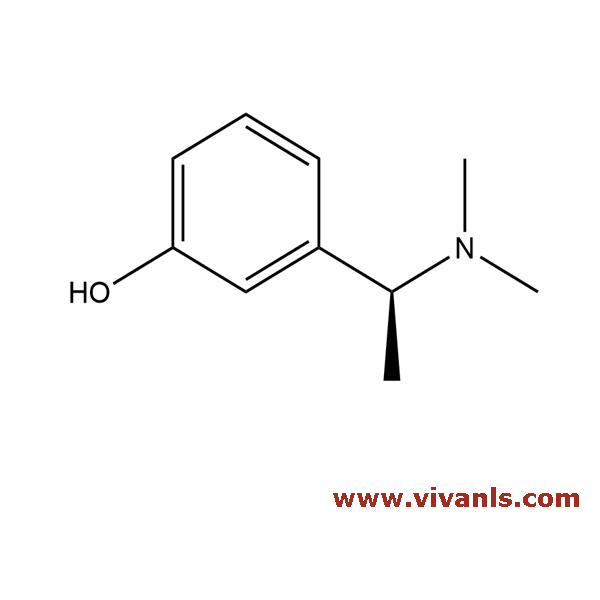
VLIM-00381
139306-10-8
C₁₀H₁₅NO
165.23

VLIM-00382
1230021-28-9
C₁₃H₂₀N₂O₂
236.31

VLIM-00383
112725-89-0
C₇H₈N₂O₃
168.15

VLIM-00384
284462-37-9
C₁₃H₁₃N₃O₂
243.26