VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00361
1199574-70-3
C₂₁H₂₉NO₃
'343.46

VLIM-00362
4335-77-7
C₁₄H₁₈O₃
'234.29

VLIM-00363
1422619-13-3
C₂₂H₂₅FN₃O₆S . 1/2 Ca
478.51
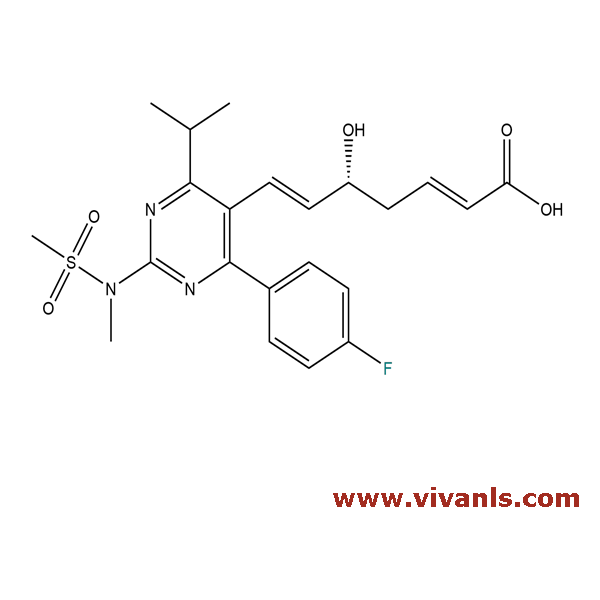
VLIM-00364
1422954-12-8
C₂₂H₂₅FN₃O₅S
462.51

VLIM-00365
1094100-06-7
C₂₂H₂₇FN₃O₆S
480.53

VLIM-00366
1258428-71-5
C₂₉H₂₆ClNO₂
455.98

VLIM-00367
909849-96-3
C₃₅H₃₆ClNO₄S
602.19

VLIM-00368
370094-12-5
C₂₂H₂₆N₂O₅S
430.52

VLIM-00369
358629-47-7
C₈H₁₂N₂O₂
168.19

VLIM-00370
74056-26-1
C₂₆H₂₉NO • HCl
407.98
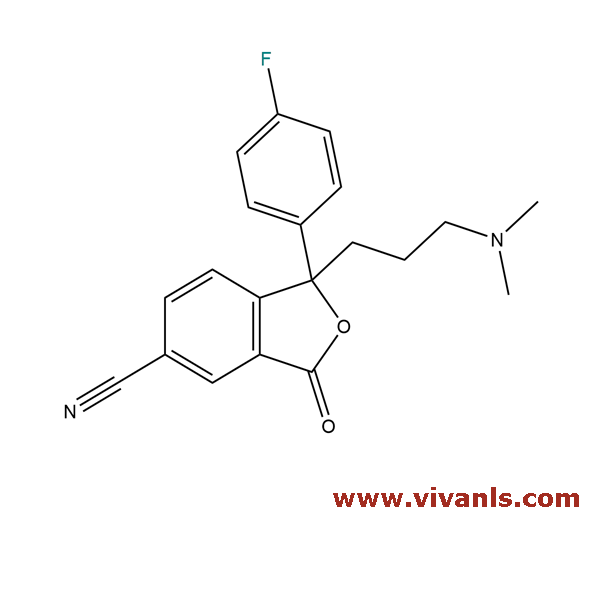
VLIM-00371
372941-54-3
C₂₀H₁₉FN₂O₂
338.38

VLIM-00372
1797986-18-5
C₄₈H₅₆Cl₄N₆O₄
922.81