VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
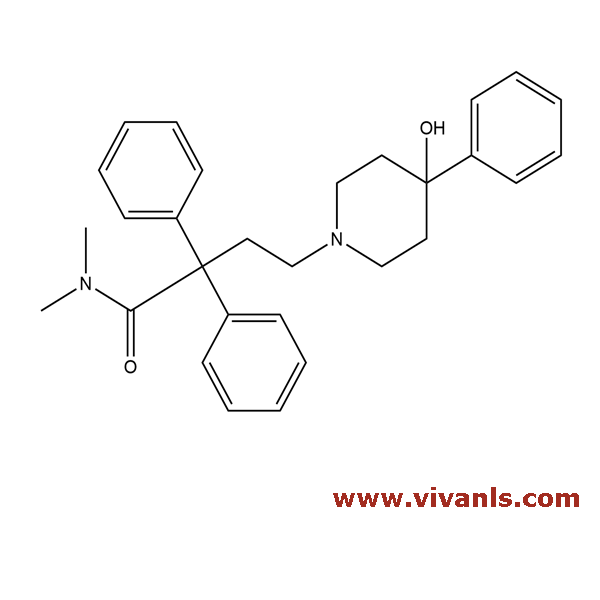
VLIM-00398
37743-41-2
C₂₉H₃₄N₂O₂
442.59
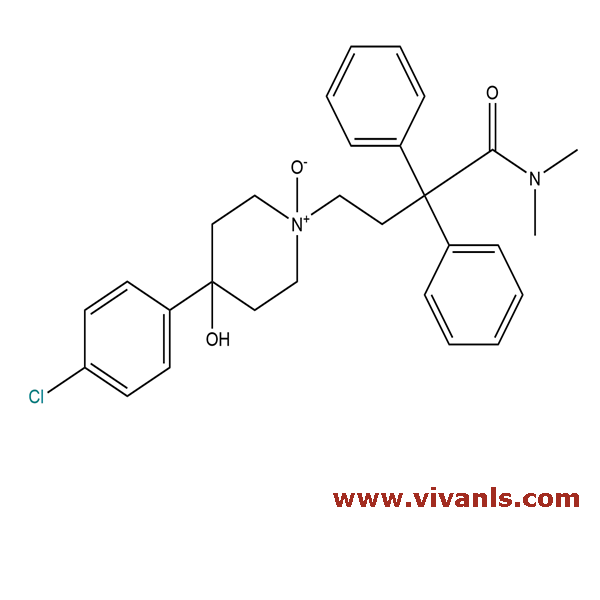
VLIM-00399
106900-12-3
C₂₉H₃₃ClN₂O₃
493.04
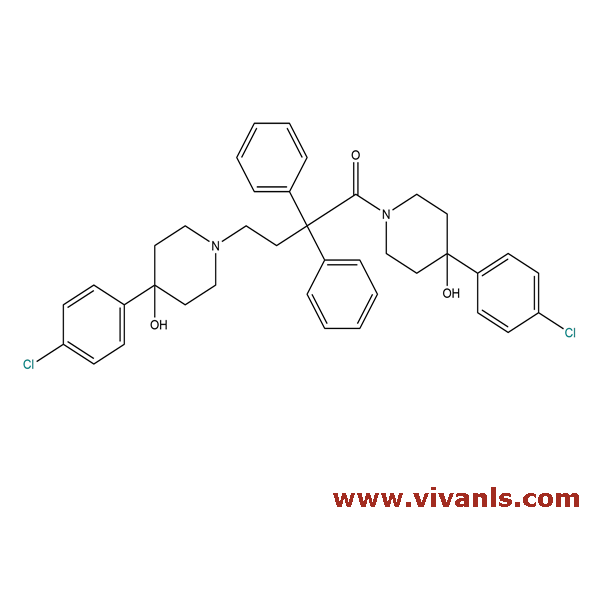
VLIM-00400
1426322-82-8
C₃₈H₄₀Cl₂N₂O₃
643.64

VLIM-00401
109572-89-6
C₂₉H₃₃ClN₂O₃
493.04

VLIM-00402
1391052-94-0
C₃₅H₃₇ClN₂O₂
553.14

VLIM-00403
117707-40-1
C₁₇H₁₈FN₃O₄
347.34

VLIM-00404
176219-04-8
C₁₇H₁₉N₃O₄S
361.42

VLIM-00405
1391053-95-4
C₂₃H₃₂N₆O₄S
488.6

VLIM-00406
82419-52-1
C₁₇H₁₈FN₃O₄
347.34

VLIM-00407
949141-22-4
C₂₁H₂₃NO₃ • HCl
373.87

VLIM-00408
103222-12-4
C₁₅H₁₆FN₃O₃
'305.30
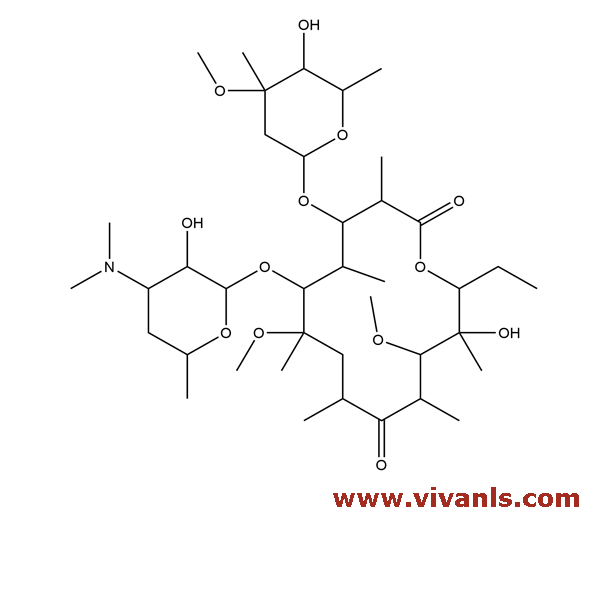
VLIM-00409
81103-14-2
C₃₉H₇₁NO₁₃
761.98