VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
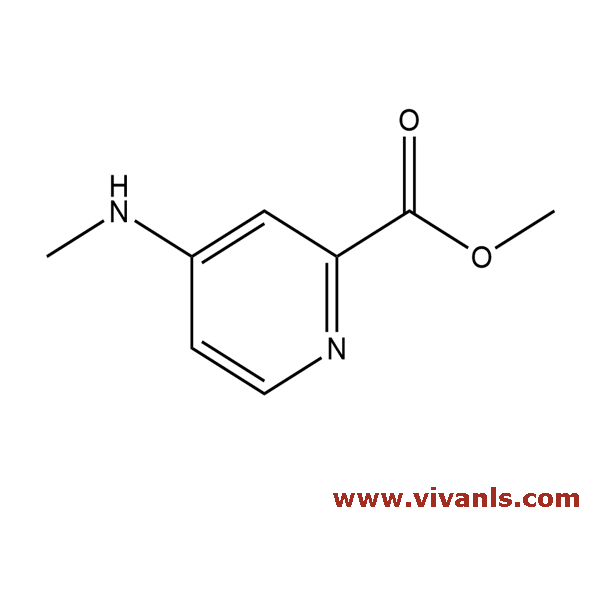
VLIM-00385
2204442-52-2
C₂₇H₂₃N₅O₆
513.5

VLIM-00386
155172-12-6
C₁₇H₁₉N₃O
281.35

VLIM-00387
73-40-5
C₅H₅N₅O
151.13

VLIM-00388
102728-64-3
C₁₀H₁₃N₅O₄
267.24

VLIM-00389
2004659-84-9
C₁₄H₁₃N₃O₃
271.27

VLIM-00390
871555-75-8
C₁₄H₉ClF₃NO₂
315.67

VLIM-00392
130606-60-9
C₂₁H₂₅ClN₂O₄S
436.95

VLIM-00393
1094598-75-0
C₂₂H₃₀N₆O₅S
490.58

VLIM-00394
139755-91-2
C₂₀H₂₆N₆O₄S
446.5

VLIM-00395
39512-49-7
C₁₁H₁₄ClNO
211.69

VLIM-00396
NA
C₄₇H₅₃BrClN₃O₃
743.3

VLIM-00397
61299-42-1
C₂₉H₃₁ClN₂O
459.02