VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00773
5111-65-9
C₁₁H₉BrO
237.09
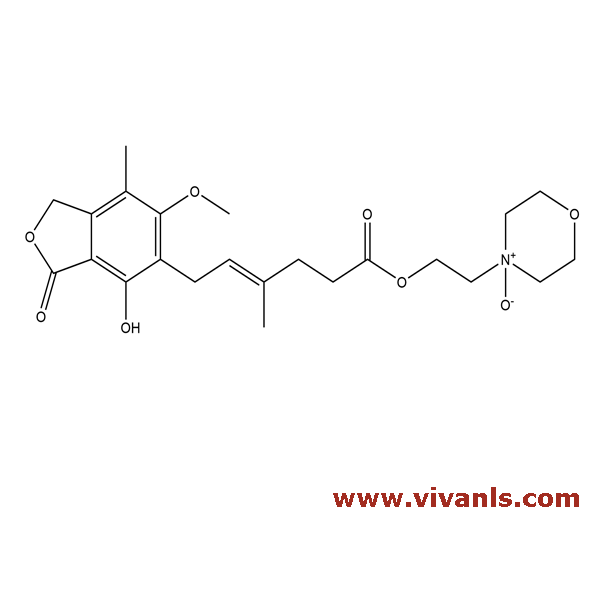
VLIM-00774
224052-51-1
C₂₃H₃₁NO₈
449.49
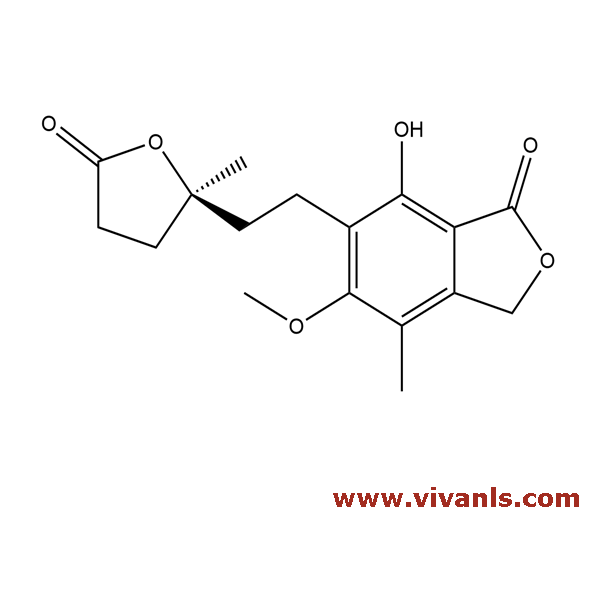
VLIM-00775
79081-87-1
C₁₇H₂₀O₆
320.34

VLIM-00776
1322681-37-7
C₂₄H₃₃NO₇
447.52

VLIM-00777
31858-66-9
C₁₈H₂₂O₆
334.36
 -1664256488.png)
VLIM-00778
100-75-4
C₅H₁₀N₂O
114.15
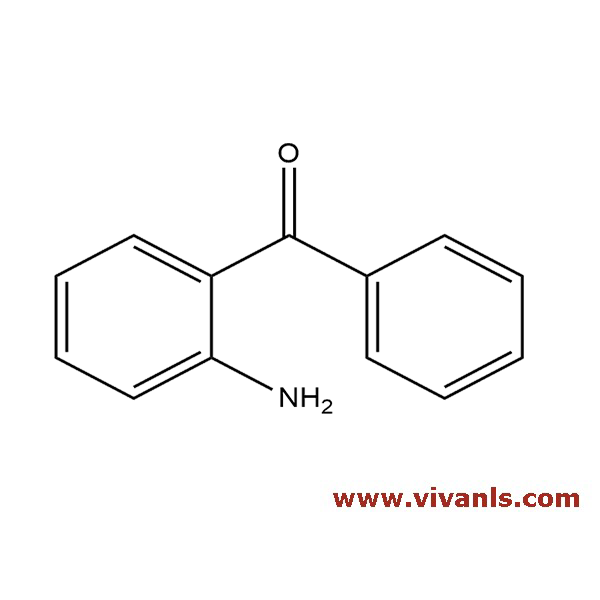
VLIM-00779
2835-77-0
C₁₅H₁₃ClN₂O₂
288.73

VLIM-00780
26159-35-3
C₁₅H₁₆O₃
244.29

VLIM-00781
1238210-10-0
C₈H₈FN₃O₄S
261.23

VLIM-00782
59-89-2
C₄H₈N₂O₂
116.12

VLIM-00783
10595-95-6
C₃H₈N₂O
88.11

VLIM-00784
614-21-1
C₈H₇NO₃
165.15