VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00797
1415566-28-7
C₁₉H₁₉N₃O₅S
401.4

VLIM-00798
1365267-35-1
C₂₉H₃₂N₆O₉
608.6

VLIM-00799
1429334-00-8
C₁₆H₁₉N₃O₅
333.34
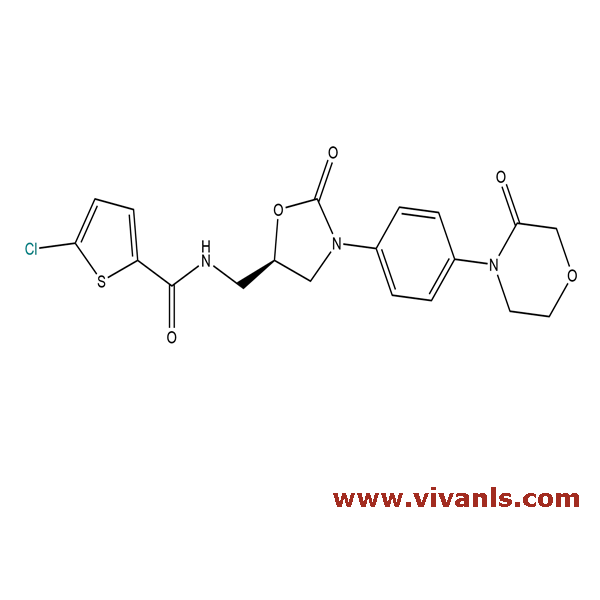
VLIM-00800
865479-71-6
C₁₉H₁₈ClN₃O₅S
435.9

VLIM-00801
4803-57-0
C₁₇H₁₇NO₃
283.32
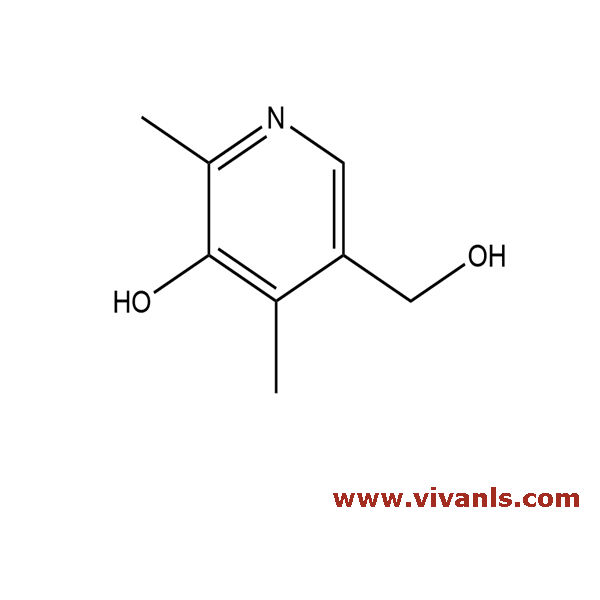
VLIM-00802
148-51-6
C₈H₁₂ClNO₂
189.64
phenol-1664363005.png)
VLIM-00803
539-15-1
C₁₀H₁₅NO
165.23

VLIM-00804
331943-04-5
C₁₇H₁₄F₃N₃O₂S
381.4

VLIM-00805
1346600-38-1
C₁₆H₂₃NO
245.36

VLIM-00806
1346601-55-5
C₁₆H₂₃NO₂
261.36

VLIM-00807
2119583-24-1
C₂₉H₂₄F₃N₇O
543.5
-Isomer-1664365344.png)
VLIM-00809
149709-63-7
C₂₄H₂₉NO₅
411.5