VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00822
24065-33-6
C₅H₃ClO₂S
162.59

VLIM-00823
581-49-7
C₁₀H₁₂N₂
160.22

VLIM-00824
2304439-07-2
C₁₃H₁₉N₃O₃ . 2HCl
338.3

VLIM-00825
135308-74-6
C₁₅H₂₃NO₂
249.35

VLIM-00826
1021933-95-8
C₁₆H₂₅NO₃
279.37

VLIM-00827
1531-23-3
C₁₇H₂₃NO
257.37

VLIM-00828
125995-03-1
C₃₃H₃₃FN₂O₄
540.62
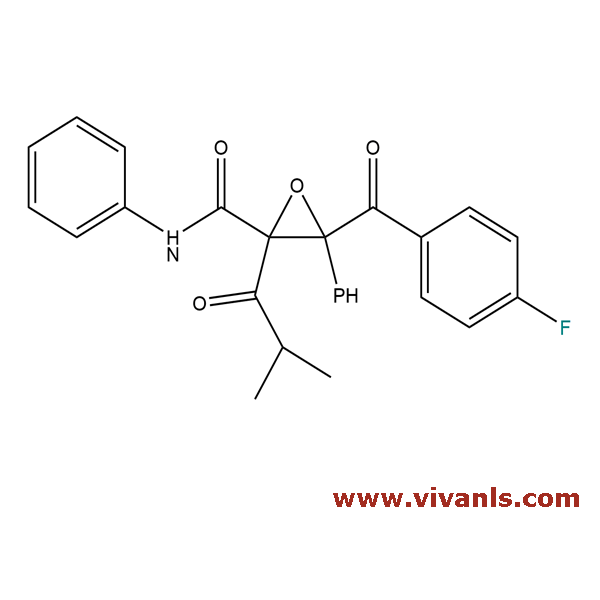
VLIM-00829
148146-51-4
C₂₆H₂₂FNO₄
431.46
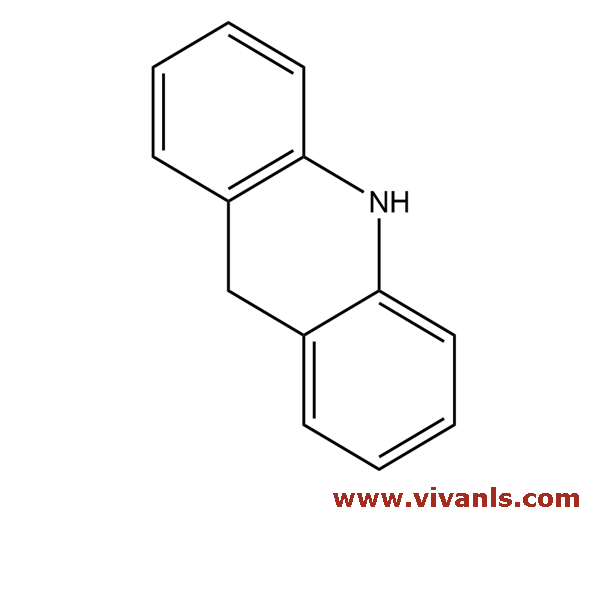
VLIM-00831
494-19-9
C₁₄H₁₃N
195.26

VLIM-00832
86393-33-1
C₁₃H₉ClFNO₃
281.67

VLIM-00833
52-21-1
C₂₃H₃₀O₆
402.48

VLIM-00835
119-36-8
C₈H₈O₃
152.15