VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
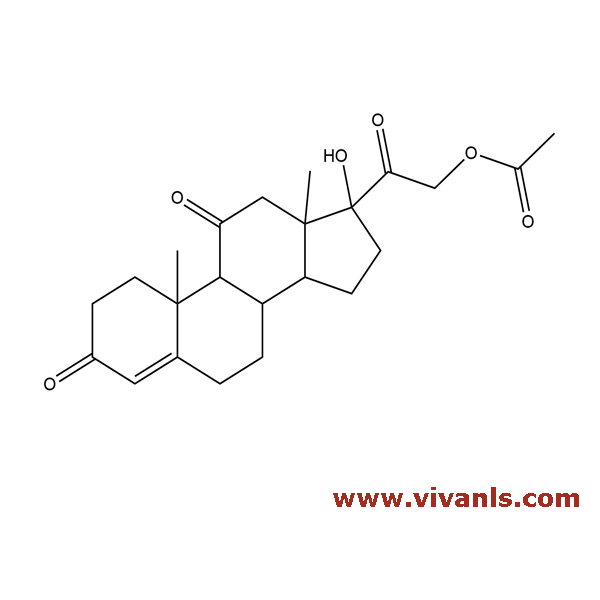
VLIM-00873
50-04-4
C₂₃H₃₀O₆
402.5
-3-((tert-Butoxycarbonyl)amino)-4-(2,4-difluorophenyl)butanoic Acid-1666075100.png)
VLIM-00874
851307-12-5
C₁₅H₁₉F₂NO₄
315.31

VLIM-00875
2346620-55-9
C₁₆H₂₃N₃O₇
369.40
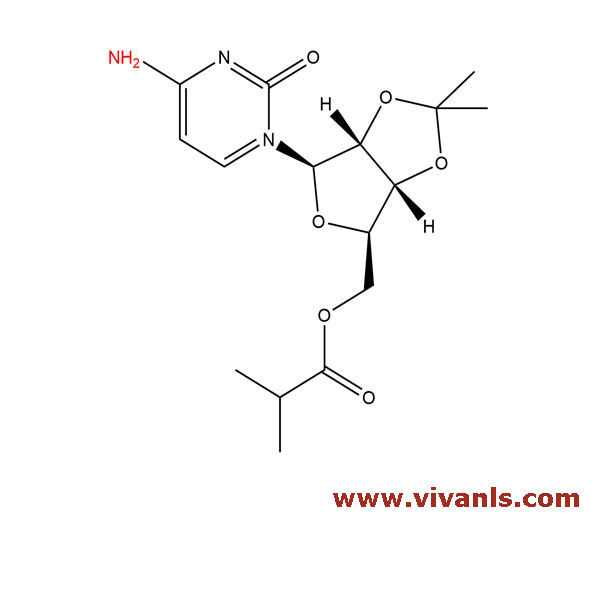
VLIM-00876
1686124-74-2
C₁₆H₂₃N₃O₆
353.37
-3-((tert-butoxycarbonyl)amino)-4-(2,5-difluorophenyl)butanoic acid-1666076153.png)
VLIM-00877
486459-98-7
C₁₅H₁₉F₂NO₄
315.32
-1666077035.png)
VLIM-00878
714269-57-5
C₂₂H₂₇ClO₇
438.9

VLIM-00879
330838-52-3
C₈H₁₃NO₂
155.19

VLIM-00880
1079990-21-8
C₅H₃FN₂O₃
158.09
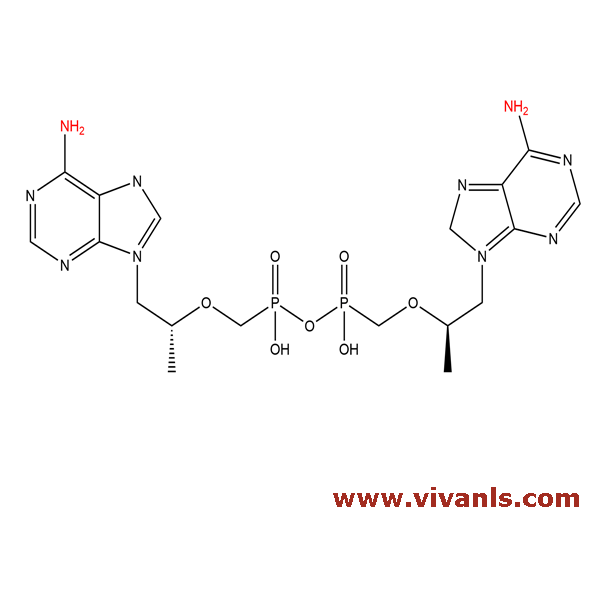
VLIM-00881
1607007-18-0
C₁₈H₂₆N₁₀O₇P₂
556.41
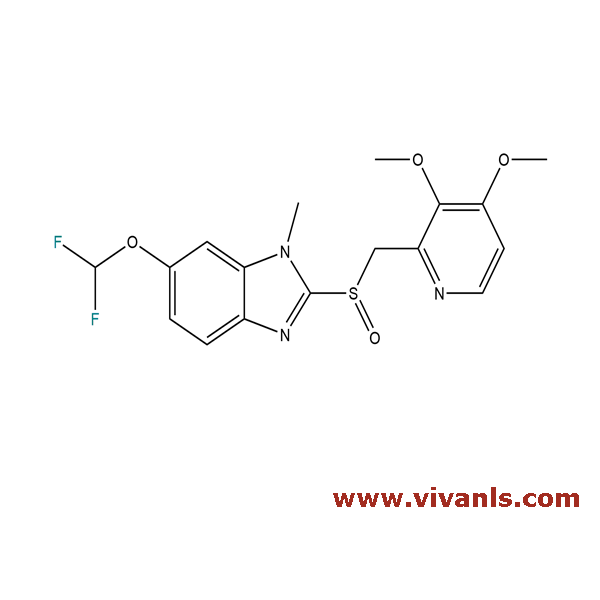
VLIM-00882
721924-06-7
C₁₇H₁₇F₂N₃O₄S
397.4

VLIM-00883
624742-53-6
C₁₇H₁₇F₂N₃O₄S
397.40

VLIM-00884
97963-62-7
C₈H₆F₂N₂OS
216.21