VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00936
120-20-7
C₁₀H₁₅NO₂
181.24

VLIM-00937
611-64-3
C₁₄H₁₁N
193.24

VLIM-00938
267668-71-3
C₁₄H₁₉ClN₆O
322.8

VLIM-00939
6258-60-2
C₈H₁₀OS
154.23

VLIM-00940
97145-88-5
C₄H₁₀ClN.HCl
144.04
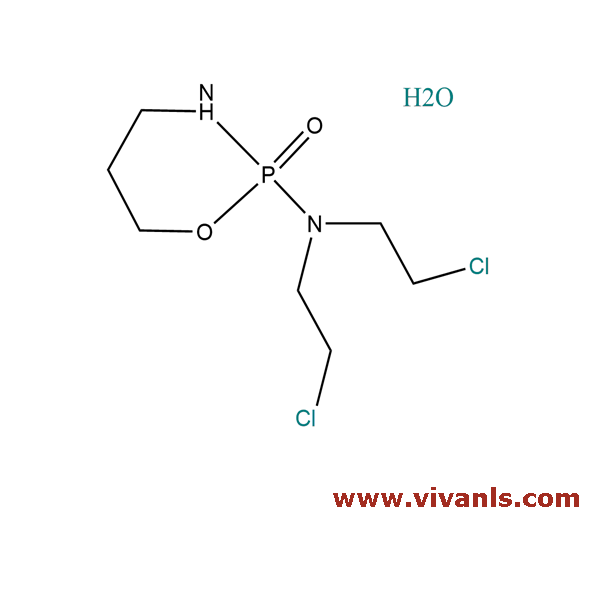
VLIM-00941
6055-19-2
C₇H₁₅Cl₂N₂O₂P · H₂O
279.1

VLIM-00942
90-15-3
C₁₀H₈O
144.17

VLIM-00943
139755-82-1
C₂₁H₂₈N₆O₄S
460.55

VLIM-00944
1049718-72-0
C₁₃H₁₉Cl₂NO
276.2

VLIM-00945
1049718-43-5
C₁₃H₁₉BrNOCl
320.65
-1-(3,4-dichlorophenyl)propan-1-one-1682506733.png)
VLIM-00946
1193779-34-8
C₁₃H₁₇Cl₂NO
274.19
propane-1,2-dione-1682506846.png)
VLIM-00947
10557-17-2
C₉H₇ClO₂
182.6