VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
-1-phenylpropan-1-one-1682507140.png)
VLIM-00948
34509-36-9
C₁₃H₁₉NO
205.3

VLIM-00949
535-80-8
ClC₆H₄CO₂H
156.57

VLIM-00950
175481-38-6
C₁₂H₁₆N₂O₃
236.27
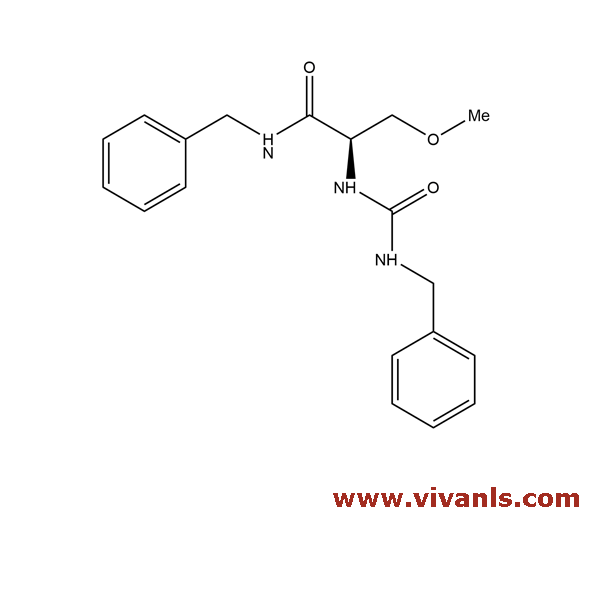
VLIM-00951
2295925-90-3
C₁₉H₂₃N₃O₃
341.4

VLIM-00952
588-46-5
C₉H₁₁NO
149.19

VLIM-00953
99-96-7
C₇H₆O₃
138.12
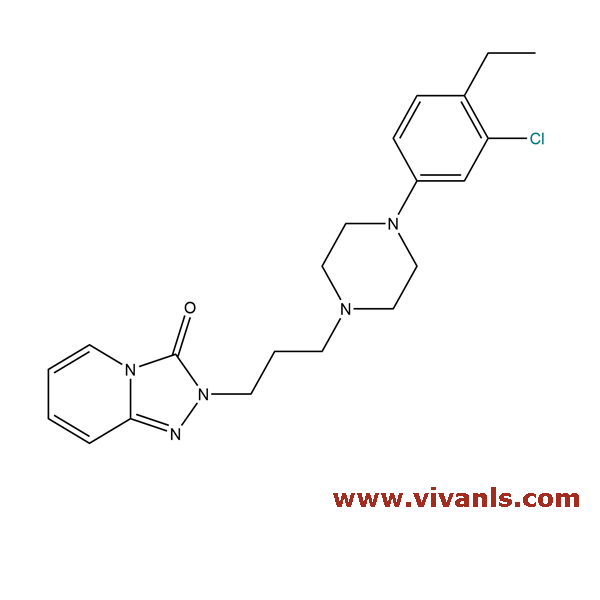
VLIM-00954
1346599-35-6
C₂₁H₂₆ClN₅O
399.92

VLIM-00955
16339-07-4
C₅H₁₁N₃O
129.16
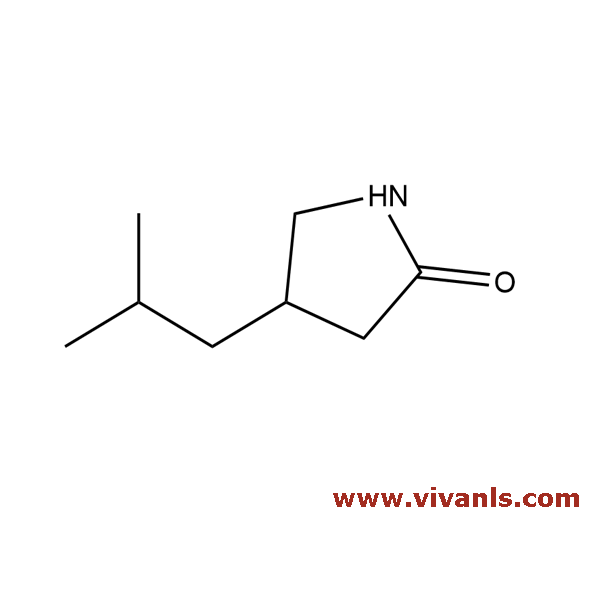
VLIM-00956
61312-87-6
C₈H₁₅NO
141.21

VLIM-00957
52413-42-0
C₁₉H₂₃N₃O
309.4

VLIM-00958
131926-99-3
C₁₆H₁₄F₃N₃O₃S
385.36

VLIM-00959
915376-75-9
C₁₅H₂₄N₂O₂.C₂H₂O₄
354.4