VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00972
167074-97-7
C₄₃H₅₁NO₁₄
805.86

VLIM-00973
1125-99-1
C₁₀H₁₇N
151.25
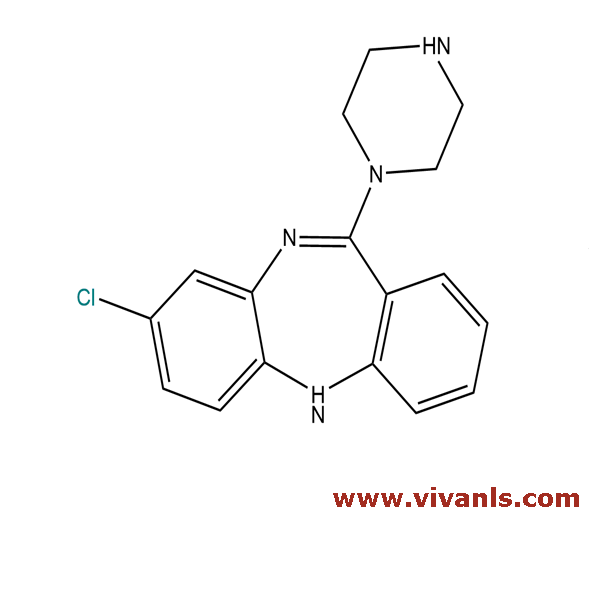
VLIM-00974
6104-71-8
C₁₇H₁₇ClN₄
312.8

VLIM-00975
3034-38-6
C₃H₃N₃O₂
113.07
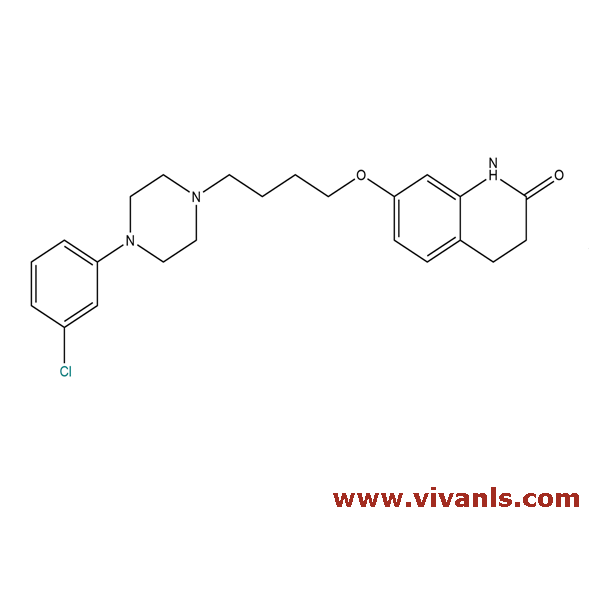
VLIM-00976
203395-82-8
C₂₃H₂₈ClN₃O₂
413.94

VLIM-00977
203395-81-7
C₂₃H₂₈ClN₃O₂
413.94

VLIM-00978
119532-26-2
C₁₀H₁₃Cl₃N₂
267.58
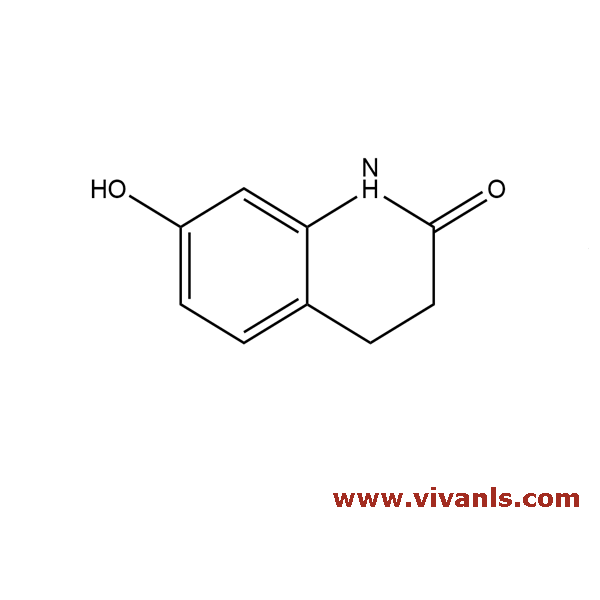
VLIM-00979
22246-18-0
C₉H₉NO₂
163.17

VLIM-00980
52589-12-5
C₄₁H₆₄O₁₅
796.96
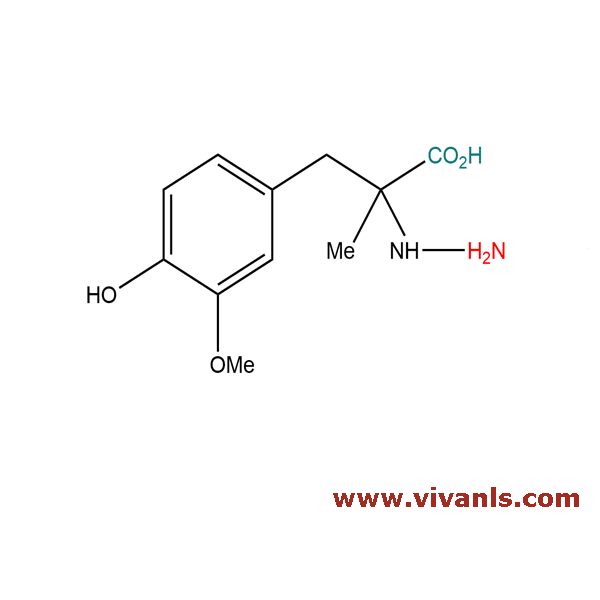
VLIM-00981
85933-19-3
C₁₁H₁₆N₂O₄
240.26

VLIM-00982
1466-67-7
C₁₅H₁₆N₂O
240.3

VLIM-00983
1318777-54-6
C₁₄H₁₈N₂O₄
278.3