VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00153
147118-40-9
C₂₃H₃₀FN₃O₆S
495.56
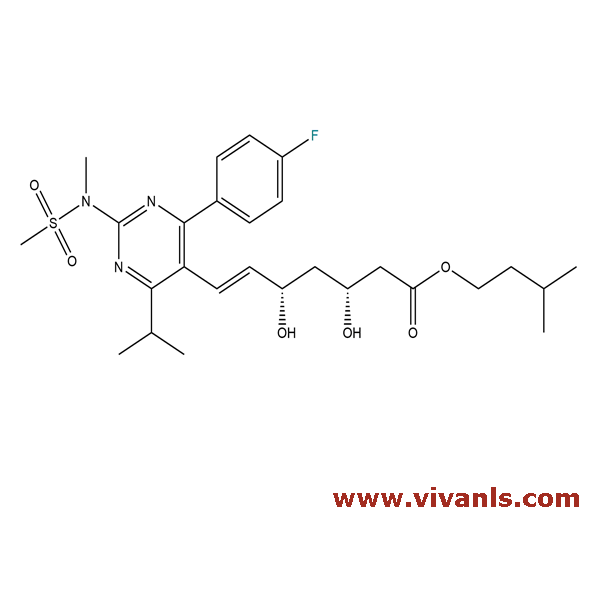
VLIM-00154
1197348-98-3
C₂₇H₃₈FN₃O₆S
551.67

VLIM-00155
103-74-2
C₇H₉NO
123.15

VLIM-00156
100-69-6
C₇H₇N
105.14

VLIM-00157
175870-21-0
C₂₅H₃₀F₃N₃O₄
493.52

VLIM-00158
887769-34-8
C₃₆H₃₉N₃O₆.HCl
646.19

VLIM-00159
2688-84-8
C₁₂H₁₁NO
185.22

VLIM-00160
5422-92-4
C₁₂H₁₀N₂O₃
230.22

VLIM-00161
372941-54-3
C₂₀H₁₉FN₂O₂
338.37
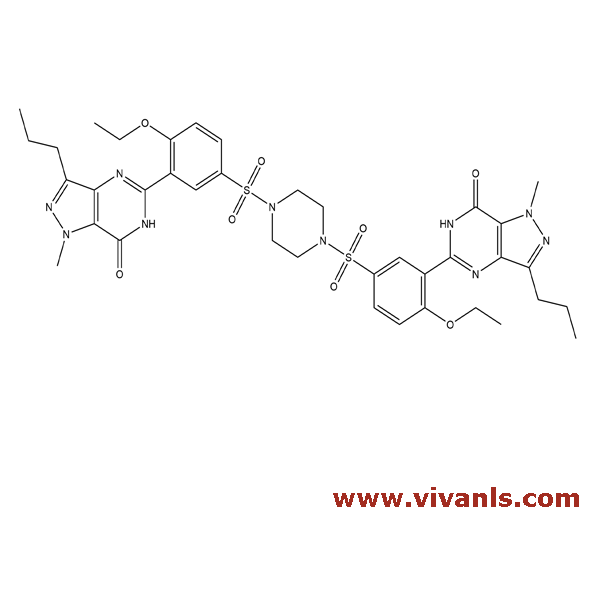
VLIM-00162
1346602-67-2
C₂₁H₂₉N₅O₆S
479.55

VLIM-00163
1106685-84-0
C₂₂H₂₂N₂O₅
394.4

VLIM-00164
1046118-40-4
C₃₃H₃₃FN₂O₆
572.62