VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
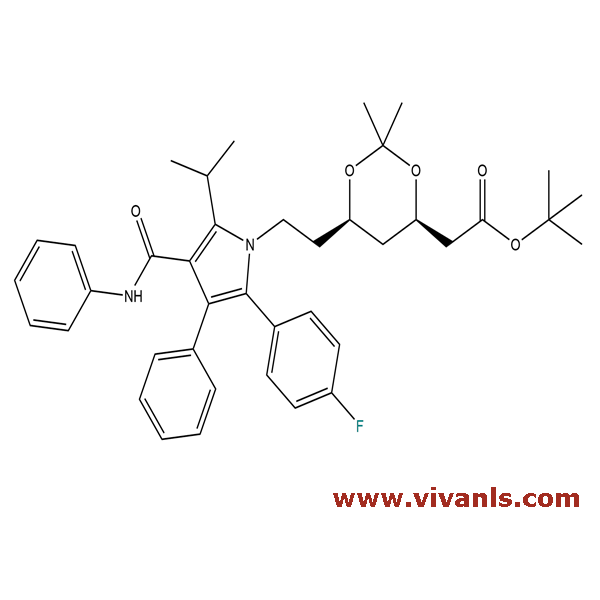
VLIM-00189
125971-95-1
C₄₀H₄₇FN₂O₅
654.81

VLIM-00190
84680-54-6
C₁₈H₂₈N₂O₇
384.42
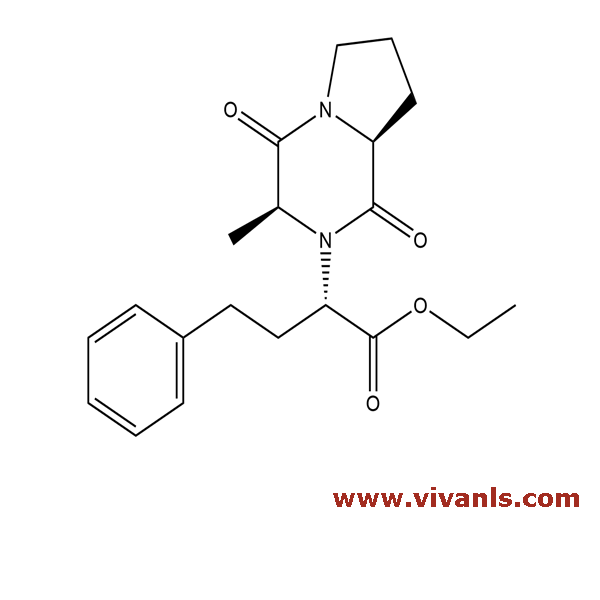
VLIM-00191
115729-52-7
C₂₀H₂₆N₂O₄
358.43

VLIM-00192
2292151-89-2
C₂₆H₃₂N₂O₅. C₄H₄O₄
568.61

VLIM-00193
76391-34-9
C₂₂H₃₂N₂O₅. C₄H₄O₄
404.51 116.07
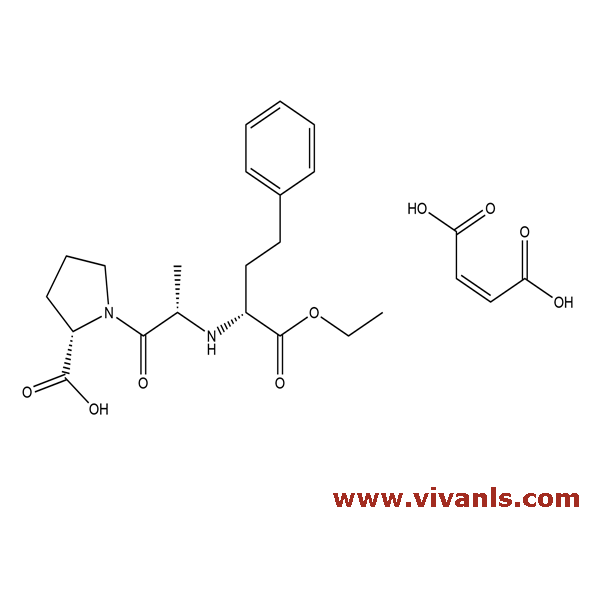
VLIM-00194
1356932-13-2
C₂₀H₂₈N₂O₅. C₄H₄O₄
376.45 116.07

VLIM-00195
82717-96-2
C₁₅H₂₁NO₄
279.33

VLIM-00196
1268997-70-1
C₂₂H₂₁ClN₄O₄S
472.95

VLIM-00197
1807733-95-4
C₂₉H₂₄ClFN₄O₄S
579.04

VLIM-00198
697299-82-4
C₂₆H₂₀ClFN₄O₂
474.91

VLIM-00199
231278-84-5
C₂₆H₁₇ClFN₃O₄
489.89

VLIM-00200
461-58-5
C₂H₄N₄
84.08