VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00089
667465-00-1
C₉H₁₄O₄
186.2

VLIM-00090
137234-88-9
C₆H₇FN₂
126.13

VLIM-00091
5872-08-02 00:00:00
C₁₀H₁₆F₂O₄S
232.3

VLIM-00092
328-90-5
C₈H₅F₃O₃
206.12

VLIM-00093
63775-95-1
C₆₁H₁₀₉N₁₁O₁₂
1188.58

VLIM-00094
59787-61-0
C₆₂H₁₁₁N₁₁O₁₃
1218.61

VLIM-00095
75330-75-5
C₂₄H₃₆O₅
404.54

VLIM-00096
79952-44-6
C₂₄H₃₆O₅
404.54
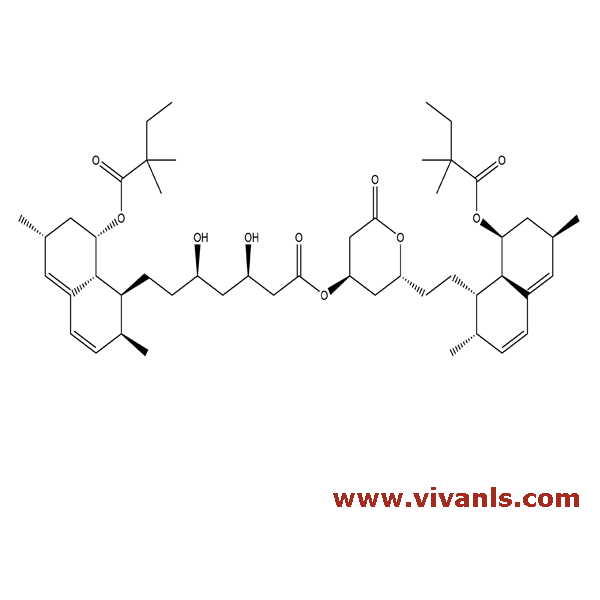
VLIM-00097
476305-24-5
C₅₀H₇₆O₁₀
837.13

VLIM-00098
1449248-72-9
C₂₅H₃₆O₅
416.55
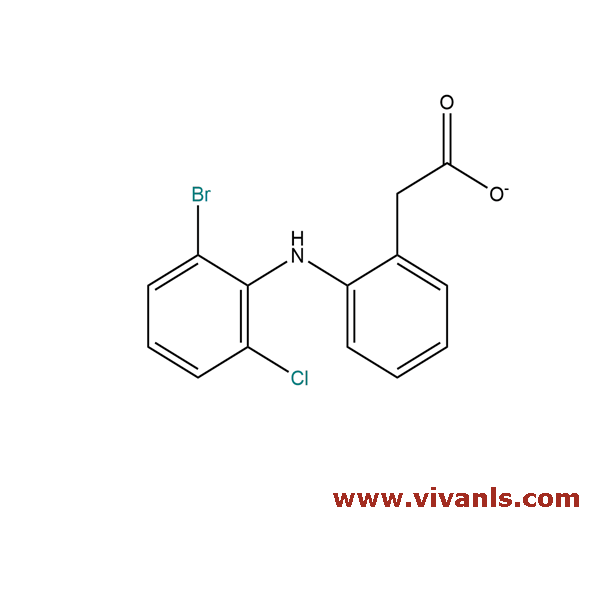
VLIM-00099
127792-45-4
C₁₄H₁₁BrClNO₂
340.59

VLIM-00100
59-48-3
C₈H₇NO
133.14