VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00064
557-48-2
C₂₇H₄₀O₃
412.6

VLIM-00066
404844-03-7
C₂₅H₂₅N₅O₄S
491.56

VLIM-00067
1798014-51-3
C₂₀H₂₅NO₄
343.423
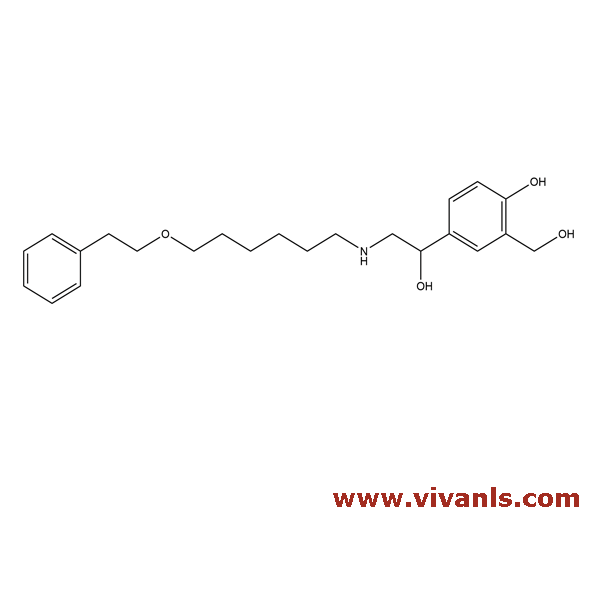
VLIM-00068
94749-02-07
C₂₃H₃₃NO₄
387.51

VLIM-00069
1391052-04-2
C₃₄H₄₇NO₇
581.75
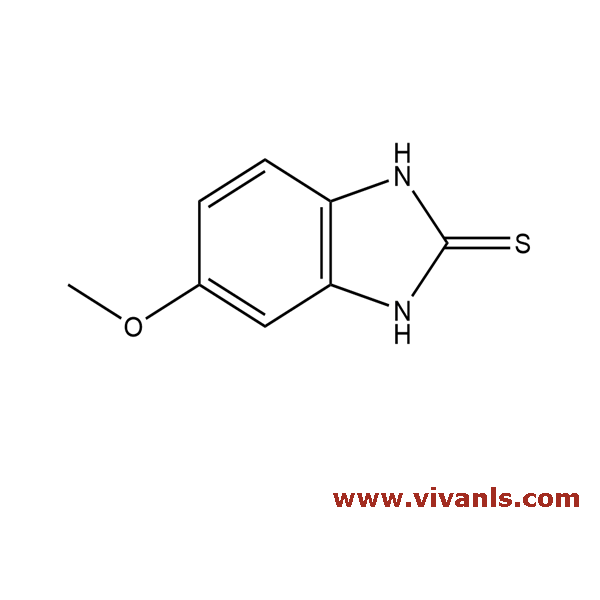
VLIM-00070
37052-78-1
C₁₇H₁₉N₃O₄S
361.42

VLIM-00071
110374-16-8
C₁₇H₁₉N₃O₄S
361.42
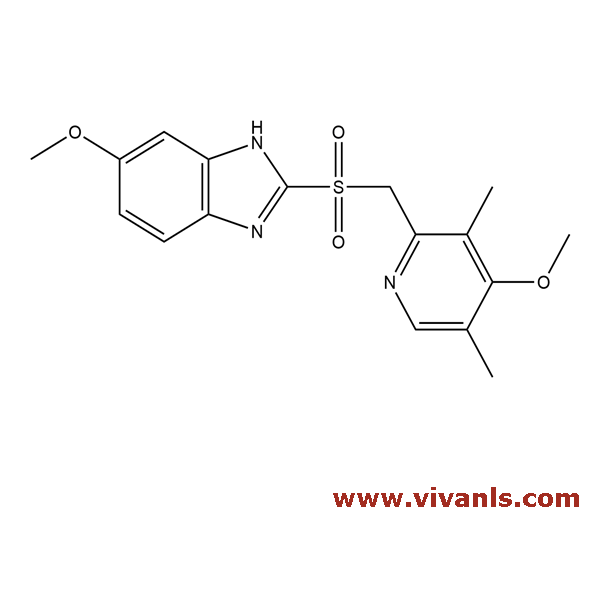
VLIM-00072
88546-55-8
C₁₇H₁₉N₃O₃S
345.42
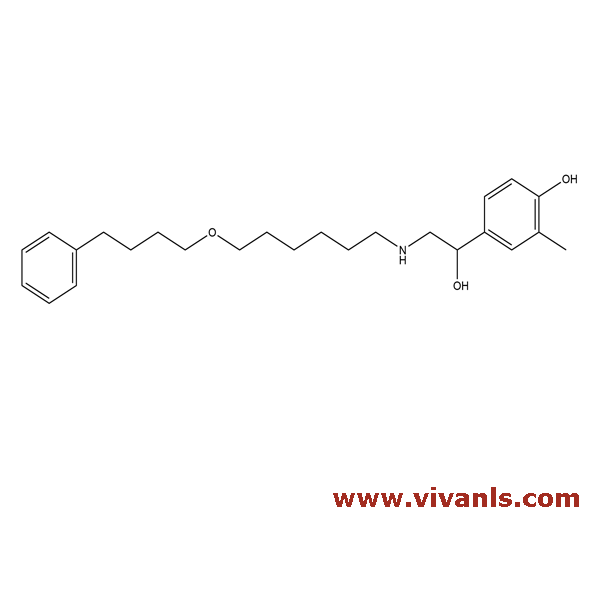
VLIM-00073
1391054-40-2
C₂₅H₃₇NO₃
399.57

VLIM-00074
150727-06-3
C₂₅H₂₄ClN₅O₄S
526.01

VLIM-00075
174227-14-6
C₂₅H₂₅N₅O₅S
507.56

VLIM-00076
1097263-60-9
C₅₂H₅₂N₁₀O₁₀S₂
1041.16