VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00039
1217214-94-2
C₁₅H₂₁F₃N₂O₂ . C₄H₄O₄
434.41

VLIM-00041
938-94-3
C₁₀H₁₂O₂
164.2

VLIM-00042
38861-78-8
C₁₂H₁₆O
176.25
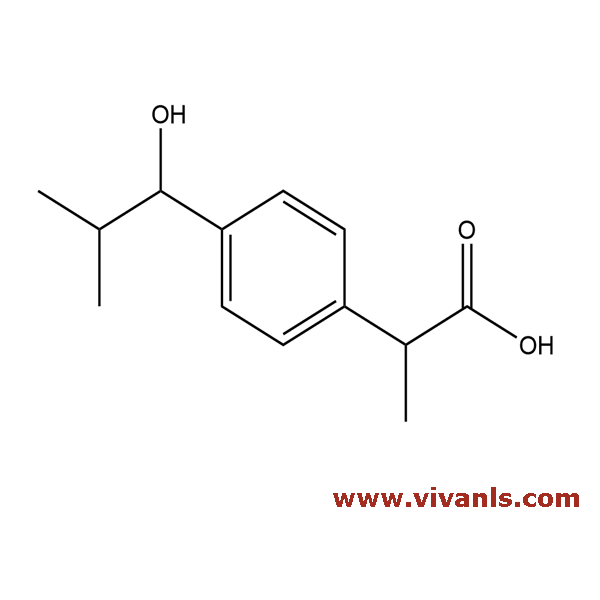
VLIM-00043
53949-53-4
C₁₃H₁₈O₃
222.28

VLIM-00044
1262894-13-2
C₁₃H₁₈O₃
222.28

VLIM-00045
65813-55-0
C₁₃H₁₆O₃
220.26

VLIM-00046
1132650-67-9
C₂₁H₃₇N₃O₅
411.53

VLIM-00047
1568181-40-7
C₉H₁₇NO₃
187.24
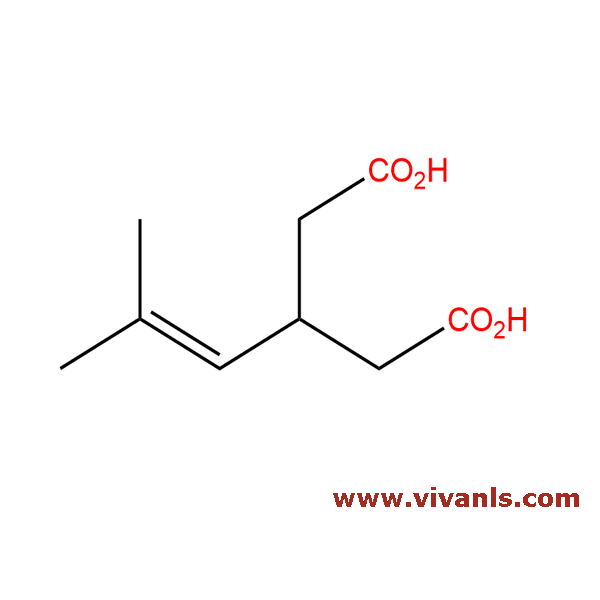
VLIM-00048
1136478-30-2
C₉H₁₆O₄
188.22
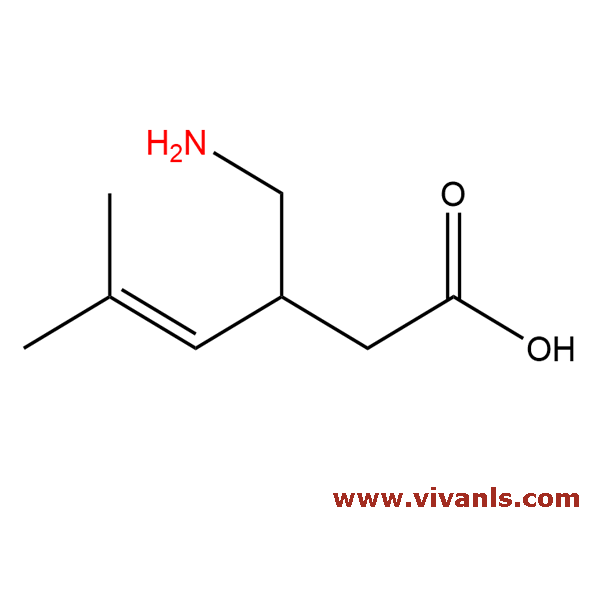
VLIM-00049
216576-74-8
C₈H₁₅NO
141.21

VLIM-00050
75143-89-4
C₈H₁₇NO₂
159.23

VLIM-00051
260435-42-5
C₁₅H₁₉N₅O
285.34